Photonic gels are colorful sensors

(Phys.org)—Materials scientists at Rice University and the Massachusetts Institute of Technology (MIT) have created very thin color-changing films that may serve as part of inexpensive sensors for food spoilage or security, multiband optical elements in laser-driven systems and even as part of high-contrast displays.
The new work led by Rice materials scientist Ned Thomas combines polymers into a unique, self-assembled metamaterial that, when exposed to ions in a solution or in the environment, changes color depending on the ions' ability to infiltrate the hydrophilic (water-loving) layers.
The research was published in the American Chemical Society journal ACS Nano.
The micron-thick material called a photonic gel, far thinner than a human hair, is so inexpensive to make that, Thomas said, "We could cover an area the size of a football field with this film for about a hundred dollars."
But for practical applications, much smaller pieces would do. "Suppose you want a food sensor," said Thomas, the William and Stephanie Sick Dean of Rice's George R. Brown School of Engineering and former chair of the Department of Materials Science and Engineering at MIT. "If it's inside a sealed package and the environment in that package changes because of contamination or aging or exposure to temperature, an inspector would see that sensor change from blue to red and know immediately the food is spoiled."

Such visual cues are good, he said, "especially when you need to look at a lot of them. And you can read these sensors with low tech, either with your own eyes or a spectrophotometer to scan things."
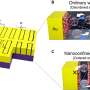
The films are made of nanoscale layers of hydrophobic polystyrene and hydrophilic poly(2-vinyl pyridine). In the liquid solution, the polymer molecules are diffused, but when the liquid is applied to a surface and the solvent evaporates, the block copolymer molecules self-assemble into a layered structure.
The polystyrene molecules clump together to keep water molecules out, while the poly(2-vinyl pyridine), P2VP for short, forms its own layers between the polystyrene. On a substrate, the layers form into a transparent stack of alternating "nano-pancakes." "The beauty of self-assembly is that it's simultaneous, all the layers forming at once," Thomas said.
The researchers exposed their films to various solutions and found different colors depending on how much solvent was taken up by the P2VP layers. For example with a chlorine/oxide/iron solution that is not readily absorbed by the P2VP, the film is transparent, Thomas said. "When we take that out, wash the film and bring in a new solution with a different ion, the color changes."
The researchers progressively turned a clear film to blue (with thiocyanate), to green (iodine), to yellow (nitrate), to orange (bromine) and finally to red (chlorine). In each case, the changes were reversible.

Thomas explained that the direct exchange of counterions from the solution to the P2VP expands those layers and creates a photonic band gap—the light equivalent of a semiconducting band gap—that allows color in a specific wavelength to be reflected. "The wavelengths in that photonic band gap are forbidden to propagate," he said, which allows the gels to be tuned to react in specific ways.
"Imagine a solid in which you create a band gap everywhere but along a 3-D path, and let's say that path is a narrowly defined region you can fabricate within this otherwise photonic material. Once you put light in that path, it is forbidden to leave because it can't enter the material, due to the band gap.
"This is called molding the flow of light," he said. "These days in photonics, people are thinking about light as though it were water. That is, you can put it in these tiny pipes. You can turn light around corners that are very sharp. You can put it where you want it, keep it from where you don't want it. The plumbing of light has been much easier than in the past, due to photonics, and in photonic crystals, due to band gaps."
More information: pubs.acs.org/doi/abs/10.1021/nn302949n
Edwin "Ned" Thomas research summary: memsweb.rice.edu/thomas/
Journal information: ACS Nano
Provided by Rice University












.jpg)