Tiny bubbles snap carbon nanotubes like twigs
What's 100 times stronger than steel, weighs one-sixth as much and can be snapped like a twig by a tiny air bubble? The answer is a carbon nanotube -- and a new study by Rice University scientists details exactly how the much-studied nanomaterials snap when subjected to ultrasonic vibrations in a liquid.
"We find that the old saying 'I will break but not bend' does not hold at the micro- and nanoscale," said Rice engineering researcher Matteo Pasquali, the lead scientist on the study, which appears this month in the Proceedings of the National Academy of Sciences.
Carbon nanotubes -- hollow tubes of pure carbon about as wide as a strand of DNA -- are one of the most-studied materials in nanotechnology. For well over a decade, scientists have used ultrasonic vibrations to separate and prepare nanotubes in the lab. In the new study, Pasquali and colleagues show how this process works -- and why it's a detriment to long nanotubes. That's important for researchers who want to make and study long nanotubes.
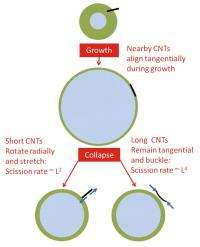
"We found that long and short nanotubes behave very differently when they are sonicated," said Pasquali, professor of chemical and biomolecular engineering and of chemistry at Rice. "Shorter nanotubes get stretched while longer nanotubes bend. Both mechanisms can lead to breaking."
Discovered more than 20 years ago, carbon nanotubes are one of the original wonder materials of nanotechnology. They are close cousins of the buckyball, the particle whose 1985 discovery at Rice helped kick off the nanotechnology revolution.
Nanotubes can be used in paintable batteries and sensors, to diagnose and treat disease, and for next-generation power cables in electrical grids. Many of the optical and material properties of nanotubes were discovered at Rice's Smalley Institute for Nanoscale Science and Technology, and the first large-scale production method for making single-wall nanotubes was discovered at Rice by the institute's namesake, the late Richard Smalley.
"Processing nanotubes in liquids is industrially important but it's quite difficult because they tend to clump together," co-author Micah Green said. "These nanotube clumps won't dissolve in common solvents, but sonication can break these clumps apart in order to separate, i.e., disperse, the nanotubes."
Newly grown nanotubes can be a thousand times longer than they are wide, and although sonication is very effective at breaking up the clumps, it also makes the nanotubes shorter. In fact, researchers have developed an equation called a "power law" that describes how dramatic this shortening will be. Scientists input the sonication power and the amount of time the sample will be sonicated, and the power law tells them the average length of the nanotubes that will be produced. The nanotubes get shorter as power and exposure time increase.
"The problem is that there are two different power laws that match with separate experimental findings, and one of them produces a length that's a good deal shorter than the other," Pasquali said. "It's not that one is correct and the other is wrong. Each has been verified experimentally, so it's a matter of understanding why. Philippe Poulin first exposed this discrepancy in the literature and brought the problem to my attention when I was visiting his lab three years ago."
To investigate this discrepancy, Pasquali and study co-authors Guido Pagani, Micah Green and Poulin set out to accurately model the interactions between the nanotubes and the sonication bubbles. Their computer model, which ran on Rice's Cray XD1 supercomputer, used a combination of fluid dynamics techniques to accurately simulate the interaction. When the team ran the simulations, they found that longer tubes behaved very differently from their shorter counterparts.
"If the nanotube is short, one end will get drawn down by the collapsing bubble so that the nanotube is aligned toward the center of the bubble," Pasquali said. "In this case, the tube doesn't bend, but rather stretches. This behavior had been previously predicted, but we also found that long nanotubes did something unexpected. The model showed how the collapsing bubble drew longer nanotubes inward from the middle, bending them and snapping them like twigs."
Pasquali said the model shows how both power laws can each be correct: One is describing a process that affects longer nanotubes and another describes a process that affects shorter ones.
"It took some flexibility to understand what was happening," Pasquali said. "But the upshot is that we have a very accurate description of what happens when nanotubes are sonicated."
More information: A copy of the PNAS paper is available at: www.pnas.org/content/early/201 … /1200013109.abstract
Journal information: Proceedings of the National Academy of Sciences
Provided by Rice University