Cells on the move
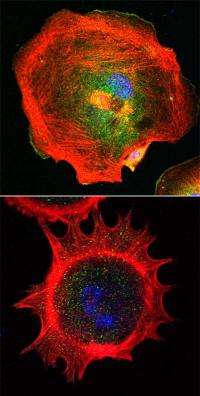
Cells on the move reach forward with lamellipodia and filopodia, cytoplasmic sheets and rods supported by branched networks or tight bundles of actin filaments. Cells without functional lamellipodia are still highly motile but lose their ability to stay on track, report researchers at the Stowers Institute for Medical Research in the April 9, 2012, online issue of the Journal of Cell Biology.
Their study provides new insight into cell motility, a complex and integrated process, which, when gone awry, can lead to various disease conditions such as cancer metastasis, birth defects, cardiovascular disease and compromised immune function.
Many cell types migrate through surrounding tissue: nerve cells reaching for their final destination; immune cells on the prowl for intruding pathogens; fibroblast called in to close wounds and stray cancer cells that have escaped the confines of the primary tumor. They all use actin filaments to push at the front by constantly remodeling their actin cytoskeleton.
"Our work demonstrates that an actin-polymerizing factor known as the Arp2/3 complex plays a critical role in the formation of the dendritic array of actin fibers that forms the structural backbone of lamellipodia and helps drive the leading edge of the cell forward," says Stowers Investigator Rong Li, Ph.D., who led the study.
When pure actin polymerizes, elongation is energetically favored over nucleation resulting in long thin filaments. The Arp2/3 complex, which localizes to lamellipodia, was thought to help build the web of actin filaments that shapes lamellipodia by initiating the branching process. "But it had been unclear whether Arp2/3 is actually required for lamellipodia formation and how it would affect cell motility," Li explains.
Unlike previous studies, which had mostly relied on RNA interference to reduce the concentration of functional Arp2/3 complex and had reached conflicting conclusions, Li and her team opted for the genetic disruption of the ARP2/3 complex to determine its function in fibroblast cell motility.
"Our study is the first one to use knock-out mice and differentiated mouse embryonic stem cells to dissect the function of Arp2/3 in fibroblast cell motility," says postdoctoral researcher and lead author Praveen Suraneni, Ph.D. Fibroblasts are very mobile and have become the standard model to study cell motility.
The Stowers researchers derived mouse embryonic stem (ES) cells from animals that lacked the gene encoding the ARPC3 subunit of the Arp2/3 complex and normal control mice and differentiated them into fibroblasts. To get a closer look at the morphology of control and mutant fibroblasts, Suraneni transferred the cells to coverslips at low density.
As soon as the cells had spread out, it became apparent that control fibroblasts sported smooth lamellipodia-like edges, while none of the mutant cells displayed the broad, veil-like protrusions reminiscent of lamellipodia. Instead, they put out spiky protrusions containing parallel actin bundles characteristic of filopodia.
To find out whether the inability to form lamellipodia impacted cells' ability to move, the researchers used a wound healing-assay to test cell motility. Wound healing is one of the main functions of fibroblasts and requires the target-oriented migration of these cells toward the site of injury. When Suraneni mimicked a wound by creating a cell-free gap between confluent layers of either normal or mutant fibroblasts, ARPC-/- cells showed a considerable delay in closing the fissure.
Time-lapse movies that tracked ARPC+/+ and ARPC-/- fibroblasts moving into the open space during the in vitro wound healing assay revealed that ARPC-/- cells moved at roughly the same speed as their wild-type counterparts. The mutant cells' failure to close the wound efficiently, was explained when Suraneni found, much to his surprise, that ARPC-/- fibroblasts showed a defect in persistent directional cell migration. In other words, they went nowhere fast.
A chemotaxis assay based on an epidermal growth factor gradient, which normal fibroblasts find irresistible, confirmed that mutant fibroblasts are unable to stay on a straight line even in the presence of a stable directional cue. "It suggests that Arp2/3 complex-mediated lamellipodia extension may be particularly favorable to cell migration processes that require strong directional persistence," says Suraneni.
Li and her team are now planning to look at different cell types derived from ARPC+/+ and ARPC-/- mouse embryonic stem cells. "Different cell types move at different speeds and respond to different cues," explains Li. "We hope to learn more about the mechanisms that drive cell motility in different contexts and whether there is a single, conserved system common to all."
Provided by Stowers Institute for Medical Research