Two-in-one imaging agents
(PhysOrg.com) -- Accurate visualization of living systems is key to the correct diagnosis and effective treatment of many diseases, as well as an improved understanding of biological processes. Magnetic resonance imaging (MRI) is a popular non-invasive visualization technique, which requires a possibly toxic contrast agent in the target tissue. Researchers have recently sought to combine MRI with confocal imaging, one of the most widely used imaging techniques in biology. For this combination to be effective, multimodal imaging agents that can function as MRI contrast agents and luminescent probes are required. Valérie C. Pierre and co-workers at the University of Minnesota report on improved magnetoluminescent systems in the European Journal of Inorganic Chemistry.
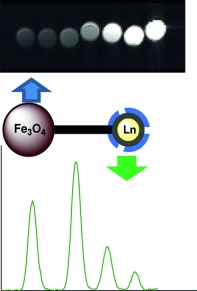
Such magnetoluminescent imaging agents consist of three components: a luminescent probe, a contrast agent, and a linker to combine the two. The use of lanthanide complexes as luminescent probes has the advantage of affording long luminescence lifetimes, which makes the system suitable for use in time-gated luminescence spectroscopy. Enhancing the absorption of the lanthanide terbium with a phenanthridine antenna provided an ideal luminescent probe. Magnetic iron oxide nanoparticles, known for their superior longitudinal and especially transverse relaxivities, were employed as the contrast agent, and a polyethylene glycol (PEG) linker was used to coat the luminescent probes onto the magnetic nanoparticles.
In addition to a precise luminescent probe and a contrast agent with excellent relaxivities, these systems are not cytotoxic, as, for example, systems held together by silica matrices. Moreover, the PEG coating is not as thick and is more water-permeable, which results in considerably improved cellular uptake and higher relaxivity.
More information: Valérie C. Pierre, Magnetoluminescent Agents for Dual MRI and Time-Gated Fluorescence Imaging, European Journal of Inorganic Chemistry, dx.doi.org/10.1002/ejic.201200045
Journal information: European Journal of Inorganic Chemistry
Provided by Wiley