A model could guide the design of artificial composites
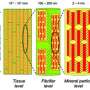
Many biomaterials such as bone, shell and mineralized tendon have a hierarchical structure that provides the material with exceptional mechanical and load-bearing properties, even though the building blocks of such structures may themselves have very poor mechanical properties. One type of structural hierarchy known as ‘self-similarity’ is ubiquitous in nature and is based on the repetition of units that are composed of biominerals and proteins, creating multi-level structures that provide enhanced strength and durability.
The number of hierarchical levels in such structures is dependent on the mineral content. Bone, for example, combines soft organic collagen material and hard crystal phases in an organized seven-level structure (see image), whereas shell is typically organized into two- or three-level structures. Little has been known, however, about what determines the number of levels in natural systems. Zuoqi Zhang at the A*STAR Institute of High Performance Computing and co-workers have now developed a theoretical, quasi-self-similar model to demonstrate why these natural biomaterials typically exhibit two to seven levels of structural hierarchy.
Previous experiments at different size scales have shown that the cooperative deformation of load-bearing biomaterials depends on their underlying hierarchical structures. The model developed by Zhang’s team, however, is the first to match these measurements of mineral and collagen deformation in bone and mineralized tendon. In the new model, each hierarchical level consists of hard, slender inclusions that form a staggered pattern within a soft matrix. These staggered microstructures then serve as inclusions in the next level. “The aspect ratio of the inclusions varies from level to level,” says Zhang.
The model showed that depending on mineral concentration, maximum toughness is obtained at a certain number of hierarchical levels and a certain hierarchical structure. Zhang notes that within the optimal structure, characteristic sizes range from tens of nanometers to hundreds of micrometers. The model also confirmed the predicted trend that the number of hierarchical levels would be highest for bone, lower for mineralized tendon, and lowest for shell. “These predictions are in agreement with experimental observations,” says Zhang.
The researchers are currently planning to use their model to guide the design and fabrication of artificial hierarchical composites in the laboratory. In addition, they are investigating the ability of hierarchical biomaterials to resist impact load. “We are trying to reveal the underlying mechanisms that may lead to acoustic cloaking composites—materials with the ability to make an object ‘invisible’ to sound,” says Zhang.
More information: Zhang, Z., et al. On optimal hierarchy of load-bearing biological materials. Proceedings of the Royal Society B 278, 519–525 (2011). rspb.royalsocietypublishing.or … content/278/1705/519
Provided by Agency for Science, Technology and Research (A*STAR)