What's the label? Helping to unravel the role of nature in biosynthetic pathways
(PhysOrg.com) -- Terpenoids are a very large and diverse class of compounds which includes certain hormones, flavors, and drugs, such as steroids, cinnamon or menthol, and antibacterials. They are found in all living organisms, but their biosynthesis is not yet fully understood. To learn more about how plants or animals make these important natural products, chemists typically turn to labeling experiments.
In these experiments, synthetic compounds are used that carry a tag or isotopic label, that is, heavy versions of atoms are chemically introduced into the precursors of biomolecules. These marked precursors are then fed to organisms. Just where these tags appear in natural terpenoids tells researchers more about how the organism made the compounds. Mevalonolactone (MVA) is an important precursor for terpenoids, and Jeroen S. Dickschat and a team of scientists from Braunschweig (Germany) have now prepared a series of labeled MVA derivatives that will help to unravel the biosynthesis of terpenoids, as they report in the European Journal of Organic Chemistry.
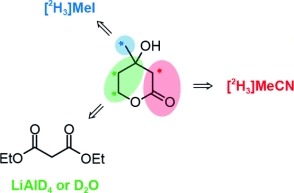
Chemists are able to follow the incorporation of isotopic labels into natural products by standard analytical methods. This is one of the most basic approaches used to determine biosynthetic pathways, as the method is only limited by the availability of the labeled compounds. Deuterated derivatives carrying a heavy version of hydrogen are a good choice for the study of terpenoids, as several steps in their biosynthesis can include rearrangements of hydrogen atoms. Deuterated MVAs have been used in the past to unravel the assembly of terpenoids; however, their availability is limited. The synthesis of such materials can be highly complex and laborious, and the isotopically labeled starting materials or reagents can be expensive. Therefore, short, efficient, and flexible routes that allow isotopic labeling at specific locations are required.
Thus, the authors set out to develop a synthetic route to deuterated MVA derivatives that allows for the independent introduction of deuterium into any position or into any combination of different positions using low-cost deuterated reagents. The team demonstrated that MVA could be labeled by using classical organic chemistry transformations, and importantly, the introduction of deuterium at any carbon atom in MVA was possible. The applicability of their route was demonstrated in the synthesis of five exemplary MVA derivatives with deuterium incorporation at different and specific locations. With the possibility to prepare new labeled MVA derivatives, scientists will now be able to address several important questions in the biosynthetic investigations of terpenoids.
More information: Jeroen S. Dickschat, Synthesis of Deuterated Mevalonolactone Isotopomers, European Journal of Organic Chemistry, Permalink to the article: dx.doi.org/10.1002/ejoc.201100188
Provided by Wiley