Conserved kinase protects cells from endosomal traffic jam
Some molecules have a single, highly specific function in physiology, while others have a much broader remit. Kinases in the IKK (inhibitor of nuclear factor κB (NF-κB) kinase) family are of the latter sort, playing a variety of roles in the immune system, cancer, and differentiation. Previous work with a related kinase, IKKε, in Drosophila has shown that this protein regulates the cytoskeleton and cell elongation in a number of contexts. It has yet to be shown, however, how it accomplishes this.
A new report by Tetsuhisa Otani in the Laboratory of Morphogenetic Signaling (Shigeo Hayashi, Group Director) and colleagues, working in collaboration with the CDB Electron Microscope and Proteomics labs, does just that. In an article published in Developmental Cell, the groups show that this factor regulates shuttling of endosomes in the tips of growing mechanosensory bristles, converting the direction of their trafficking from incoming to outward bound.
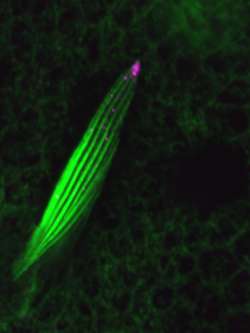
Bristles form from single cells during the fly’s pupal stage. A previous report had indicated that IKKε is expressed at the tips of these structures. Otani looked at the active, phosphorylated form of the molecule and found that it indeed accumulated at the tips of growing bristles. He looked closer at cytoskeletal organization in these structures in wildtype and IKKε mutants, and found that in the mutant bristles, actin bundles were poorly organized and frequently had failed to attach to their anchor points in the cell cortex. Transmission electron microscopy further showed that while microtubules in mutants maintained their usual orientation with the respect to the bristle, an abnormal number of vesicles had accumulated in this space, suggesting that vesicle transport was somehow affected.
An examination of vesicle markers turned up a promising candidate in Rab11, a recycling endosome protein known to be essential in bristle morphogenesis. In contrast to its tendency to cluster around the tip in control animals, Rab11 was clumped in the bristle shaft in IKKε mutants. Other endosome markers also showed aberrant localization. The effect was replicated in cultured cells, in which mutant IKKε caused Rab11 to aggregate.
What then was the link between the defects in actin bundles and endosome distribution in IKKε mutants? Looking at the timing of these events, Otani found that Rab11 accumulation precedes the misrouting of actin, and was unaffected by various mutations that disturb the bundling of actin filaments, suggesting that IKKε’s roles in Rab11 distribution and actin bundling are independent.
He next used photobleaching and time-lapse imaging to track the localization of Rab11 over time. Rab11 normally moves along the bristle, shuttling between shaft and tip in a state of dynamic equilibrium. This rate and overall directionality of this back-and-forth varied, and was at its highest during bristle elongation. Looking for a mechanism, the group examined functional relationships between IKKε, Rab11, and the Rab11 effector, Nuf, which binds to Rab11 and the Dynein light intermediate chain (a component in microtubule trafficking). What they found pointed to opposing roles for IKKε and Nuf in the trafficking of Rab11. This antagonistic effect appears to be due to the phosphorylation of Nuf at a specific amino acid (serine 225) by IKKε. Preliminary experiments using IKKε homologs in mammalian cells suggested that the function of this factor in the trafficking of recycling endosomes is conserved.
"In addition to their role as vehicles for cellular materials, endosomes also serve as a platform for various signaling activities in cell proliferation and immunity,” says Hayashi. “The identification of new downstream targets of IKKε sheds light on the roles of endosomal trafficking in morphogenesis, innate immunity, and cancer."
More information: Otani T, et al. 'IKKɛ Regulates Cell Elongation through Recycling Endosome Shuttling.' Dev Cell. 2011 Feb 15;20(2):219-32. www.ncbi.nlm.nih.gov/pubmed/21316589?dopt=Citation
Provided by RIKEN