New life form found on Earth: Deadly arsenic breathes life into organisms (Update, Video)

(PhysOrg.com) -- Evidence that the toxic element arsenic can replace the essential nutrient phosphorus in biomolecules of a naturally occurring bacterium expands the scope of the search for life beyond Earth, according to Arizona State University scientists who are part of a NASA-funded research team reporting findings in the Dec. 2 online Science Express.
It is well established that all known life requires phosphorus, usually in the form of inorganic phosphate. In recent years, however, astrobiologists, including Arizona State University professors Ariel Anbar and Paul Davies, have stepped up conversations about alternative forms of life. Anbar and Davies are coauthors of the new paper, along with ASU associate research scientist Gwyneth Gordon. The lead author is Felisa Wolfe-Simon, a former postdoctoral scientist in Anbar's research group at ASU's School of Earth and Space Exploration and Department of Chemistry and Biochemistry in the College of Liberal Arts and Sciences.
"Life as we know it requires particular chemical elements and excludes others," says Anbar, a biogeochemist and astrobiologist who directs the astrobiology program at ASU. "But are those the only options? How different could life be?" Anbar and Wolfe-Simon are among a group of researchers who are testing the limits of life's chemical requirements.
"One of the guiding principles in the search for life on other planets, and of our astrobiology program, is that we should 'follow the elements,'" says Anbar. "Felisa's study teaches us that we ought to think harder about which elements to follow."
Wolfe-Simon adds: "We took what we do know about the 'constants' in biology, specifically that life requires the six elements CHNOPS (carbon, hydrogen, nitrogen, oxygen, phosphorus and sulfur) in three components, namely DNA, proteins and fats, and used that as a basis to ask experimentally testable hypotheses even here on Earth."
From this viewpoint, rather than highlighting the conventional view of the "diversity" of life, all life on Earth is essentially identical, she says. However, the microbe the researchers have discovered can act differently.
Davies has previously speculated that forms of life different from our own, dubbed "weird life," might even exist side-by-side with known life on Earth, in a sort of "shadow biosphere." The particular idea that arsenic, which lies directly below phosphorous on the periodic table, might substitute for phosphorus in life on Earth, was proposed by Wolfe-Simon and developed into a collaboration with Davies and Anbar. Their hypothesis was published in January 2009, in a paper titled "Did nature also choose arsenic?" in the International Journal of Astrobiology.
"We not only hypothesized that biochemical systems analogous to those known today could utilize arsenate in the equivalent biological role as phosphate," notes Wolfe-Simon "but also that such organisms could have evolved on the ancient Earth and might persist in unusual environments today."
Wolfe-Simon, now a NASA astrobiology research fellow in residence at the U.S. Geological Survey, was one of the participants, along with Anbar, at a workshop titled "Tree or Forest? Searching for Alternative Forms of Life on Earth," that was organized in December 2006 by the BEYOND Center, a "cosmic think tank" at ASU.
"That's where it all began," says Davies, a cosmologist, astrobiologist, theoretical physicist and director of the BEYOND Center.
"Felisa's talk was memorable for being a concrete proposal," Davies says. "Many of the talks at the workshop discussed searching for radically alternative forms of life with suggestions of the form 'maybe something roughly like this,' or 'maybe a bit like that.' But Felisa said, quite explicitly, 'this is what we go look for.' And, she did."
"The idea was provocative, but it made good sense," notes Anbar. "Arsenic is toxic mainly because its chemical behavior is so similar to that of phosphorus. As a result, organisms have a hard time telling these elements apart. But arsenic is different enough that it doesn't work as well as phosphorus, so it gets in there and sort of gums up the works of our biochemical machinery."
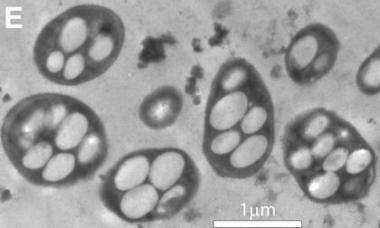
After leaving ASU, Wolfe-Simon began a collaboration with Ronald Oremland of the U.S. Geological Survey to chase down the hypothesis. Oremland was a natural choice to bring into the project because he is a world expert in arsenic microbiology. What Wolfe-Simon discovered is presented in the Science Express paper titled "A bacterium that can grow by using arsenic instead of phosphorus."
The latest discovery is all about a bacterium – strain GFAJ-1 of the Halomonadaceae family of Gammaproteobacteria – scooped from sediments of eastern California's Mono Lake, which is extremely salty with naturally high levels of arsenic.
In the laboratory, the researchers successfully grew microbes from the lake on a diet that was very lean on phosphorus, but included generous helpings of arsenic.
Key issues that the researchers needed to address were the levels of arsenic and phosphorus in the experiments and whether arsenic actually became incorporated into the organisms' vital biochemical machinery, such as DNA, proteins and the cell membranes. A variety of sophisticated laboratory techniques was used to nail down where the arsenic went, including mass spectrometry measurements by Gordon at the W.M. Keck Foundation Laboratory for Environmental Biogeochemistry at ASU.
Commenting on the significance of the discovery, Davies says: "This organism has dual capability. It can grow with either phosphorous or arsenic. That makes it very peculiar, though it falls short of being some form of truly 'alien' life belonging to a different tree of life with a separate origin. However, GFAJ-1 may be a pointer to even weirder organisms. The holy grail would be a microbe that contained no phosphorus at all."
Davies predicts that the new organism "is surely the tip of a big iceberg, and so has the potential to open up a whole new domain of microbiology."
It is not only scientists, however, who will be interested in this discovery. "Our findings are a reminder that life-as-we-know-it could be much more flexible than we generally assume or can imagine," says Wolfe-Simon, noting that because microbes are major drivers of biogeochemical cycles and disease this study may open up a whole new chapter in biology textbooks.
"Yet, this story isn't about arsenic or Mono Lake," Wolfe-Simon says. "If something here on Earth can do something so unexpected, what else can life do that we haven't seen yet? Now is the time to find out."
------------------------
Earlier story (Updated 12/2/2010 12:40pm EST):
NASA will announce later Thursday an explosive finding in the search for new life forms on Earth and beyond, a discovery that shifts what scientists consider necessary for life to exist.
While details of the study are under embargo until 2:00 pm (1900 GMT), media leaks have already established that the findings relate to a bacteria that survives and grows on arsenic, uncovered deep in a California lake.
The discovery of a bacteria that can incorporate arsenic into its DNA adds a new dimension to what has long been accepted as the building blocks of life as we know it.
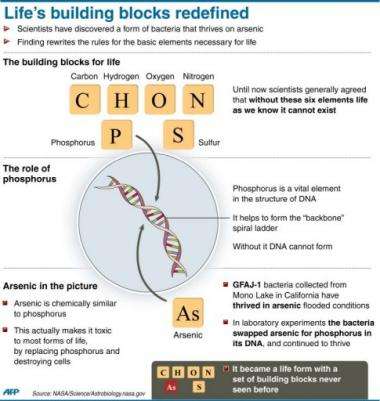
Until now, biologists have said life requires six elements, known by the acronym CHNOPS: carbon, hydrogen, nitrogen, oxygen, phosphorus and sulfur.
Those elements must take shape within three components of an organism: DNA, protein and fats.
Every life form has DNA. It is the identifier that, for example, allows humans to reproduce other humans while mice give birth to mice.
In other words, DNA is a molecule (called deoxyribonucleic acid) that gives life its biological instructions.
All known life needs phosphorus, which serves as the backbone for these nucleic acids that make up life's genetic material.
Arsenic is right below phosphorus on the periodic table, and the two are very similar.
However arsenic is different enough that it cannot substitute for phosphorus, and typically slows down an organism enough that it kills it, hence why it is considered poison.
But one of the speakers scheduled to appear at the press conference later Thursday, Felisa Wolfe-Simon, wondered if it had always been that way.
In 2009 she co-authored a paper called "Did nature also choose arsenic?"
In that paper, she wrote: "We hypothesize that ancient biochemical systems, analogous to but distinct from those known today, could have utilized arsenate in the equivalent biological role as phosphate.
"Organisms utilizing such 'weird life' biochemical pathways may have supported a 'shadow biosphere' at the time of the origin and early evolution of life on Earth or on other planets.
"Such organisms may even persist on Earth today, undetected, in unusual niches."
Of course, another condition for life to exist is adaptability. All life must have adapted, or adapt now or in the future, in order to survive.
Previous scientific discoveries have found examples of life forms existing on Earth in extreme heat, cold, acidic environments and airless underground pockets.
Scientists have also already found examples of bacteria that use arsenic the way humans use food and oxygen for energy.
So it is possible that this odd bacteria was only doing what it had to in order to survive in a lake with unusually high levels of arsenic, salt and other minerals.
NASA's vague announcement earlier this week of a press conference on "an astrobiology finding that will impact the search for evidence of extraterrestrial life" set the Internet abuzz with speculation.
Astrobiology relates to the study of life in the universe, including its origin and evolution, where it is located and how it might survive in the future.
The full details of the study will be published later today in the journal Science Express, at the same time as the NASA press conference begins.
Other speakers are to include Mary Voytek, director of the Astrobiology Program at NASA; Pamela Conrad, a NASA astrobiologist; Steven Benner, distinguished fellow at the Foundation for Applied Molecular Evolution; and James Elser, professor at Arizona State University.
Provided by Arizona State University