This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
proofread
Researchers develop novel procedure for isolating primary mouse hepatocytes with holographic acoustic tweezers
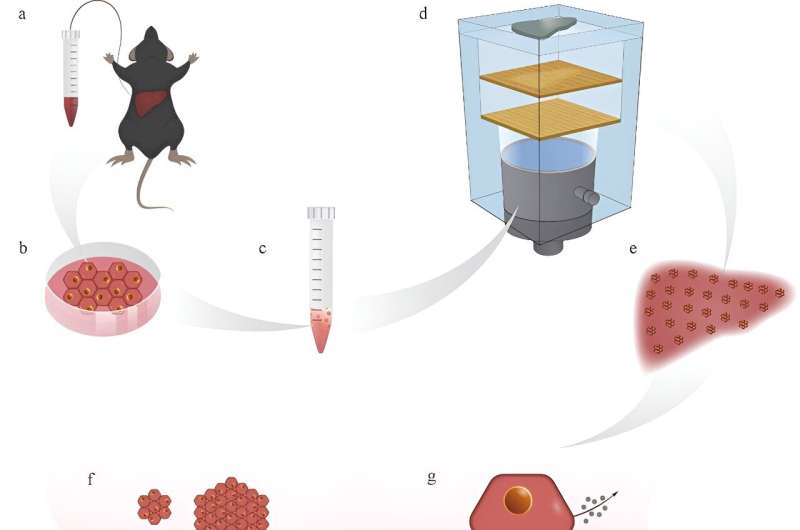
Recently, a research team established a comprehensive procedure for isolating primary mouse hepatocytes and maintaining them in long-term culture with significant amplification in a two-dimensional (2D) environment. The team was led by Prof. Li Fei and Zheng Hairong from the Shenzhen Institute of Advanced Technology (SIAT) of Chinese Academy of Sciences, along with Prof. Mao Yilei and Yang Huayu from the Peking Union Medical College (PUMC) Hospital,
The study is published in Biomaterials.
Primary cells sourced directly from organisms have become indispensable tools for cellular and molecular biology research in recent years. However, the current three-dimensional (3D) culture models suffer from drawbacks such as a limited lifespan, stringent culture requirements, complicated procedures, and restricted cell numbers, hindering their applications, especially in isolating and culturing primary hepatocytes in vitro.
Holographic acoustic tweezers (HAT) exhibit significant potential in constructing in vitro 3D models for investigating the mechanisms of cancer, testing and selecting suitable anticancer drugs, and developing new medications. However, there is a lack of in-depth research on the biological effects of HAT on primary cells.
In this study, the researchers improved the existing HAT technology and proposed a model of holographic lattice-based acoustic tweezers. They developed a group of acoustic holographic lenses to modulate the phase and amplitude of the incident sound wave simultaneously, to generate any complex-patterned acoustic holographic lattice array, such as circular, triangular, square, and liver-shaped complex acoustic holographic lattice patterns.
Subsequently, they used the acoustic radiation force of the acoustic potential well to assemble cells into specific lattice patterns, simulating the anatomical structure of the liver.
In addition, the researchers used expandable primary mouse liver cells in vitro for the first time to deeply study the impact of HAT on the biological effects of primary liver cells. The results showed that primary liver cells under the holographic lattice-based acoustic tweezers produced a large number of self-assembled liver cell spheroids. Compared with traditional 2D and 3D culture models, the diameter of liver cell spheroids significantly increased, and the functions of liver cell protein synthesis metabolism, sugar metabolism, detoxification, and more were also significantly enhanced.
This work establishes the foundation for the development of an ex vivo model through acoustical holographic lattice techniques, carrying significant implications for future 3D tissue engineering applications.
More information: Changcan Li et al, Acoustic-holography-patterned primary hepatocytes possess liver functions, Biomaterials (2024). DOI: 10.1016/j.biomaterials.2024.122691
Journal information: Biomaterials
Provided by Shenzhen Institute of Advanced Technology, Chinese Academy of Sciences