This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Silkworms help grow better organ-like tissues in labs
Biomedical engineers at Duke University have developed a silk-based, ultrathin membrane that can be used in organ-on-a-chip models to better mimic the natural environment of cells and tissues within the body. When used in a kidney organ-on-a-chip platform, the membrane helped tissues grow to recreate the functionality of both healthy and diseased kidneys.
By allowing the cells to grow closer together, this new membrane helps researchers to better control the growth and function of the key cells and tissues of any organ, enabling them to more accurately model a wide range of diseases and test therapeutics.
The research was published June 5 in the journal Science Advances.
Often no larger than a USB flash drive, organ-on-a-chip (OOC) systems have revolutionized how researchers study the underlying biology of the human body, whether it's creating dynamic models of tissue structures, studying organ functions or modeling diseases.
These platforms are designed to stimulate cell growth and differentiation in a way that best mimics the organ of interest. Researchers can even populate these tools with human stem cells to generate patient-specific organ models for pre-clinical studies.
But as the technology has evolved, problems in the chip's design have also emerged—most notably with the materials used to create the membranes that form the support structure for the specialized cells to grow on.
These membranes are typically composed of polymers that don't degrade, creating a permanent barrier between cells and tissues. While the extracellular membranes in human organs are often less than one micron thick, these polymer membranes are anywhere from 30 to 50 microns, hindering communication between cells and limiting cell growth.
"We want to handle the tissues in these chips just like a pathologist would handle biopsy samples or even living tissues from a patient, but this wasn't possible with the standard polymer membranes because the extra thickness prevented the cells from forming structures that more closely resemble tissues in the human body.
"We thought, 'Wouldn't it be nice if we could get a protein-based material that mimics the structure of these natural membranes and is thin enough for us to slice and study?'" says Samira Musah, assistant professor of biomedical engineering.
This question led Musah and George (Xingrui) Mou, a Ph.D. student in Musah's lab and first author on the paper, to silk fibroin, a protein created by silkworms that can be electronically spun into a membrane. When examined under a microscope, silk fibroin looks like spaghetti or a Jackson Pollock painting.
Made out of long, intertwining fibers, the porous material better mimics the structure of the extracellular matrix found in human organs, and it has previously been used to create scaffolds for purposes like wound healing.
"The silk fibroin allowed us to bring the membrane thickness down from 50 microns to 5 or fewer, which gets us an order of magnitude closer to what you'd see in a living organism," explained Mou.
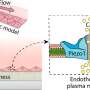
To test this new membrane, Musah and Mou applied the material to their kidney chip models. Made out of a clear plastic and roughly the size of a quarter, this OOC platform is meant to resemble a cross section of a human kidney—specifically the glomerular capillary wall, a key structure in the organ made from clusters of blood vessels that is responsible for filtering blood.
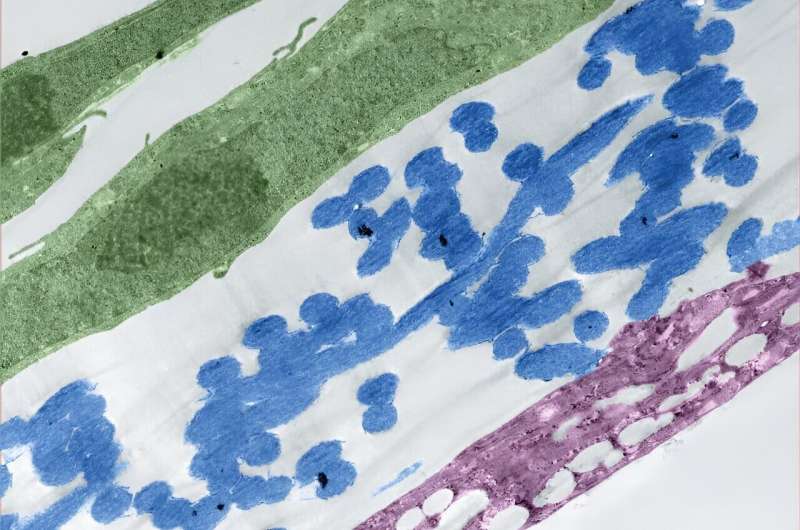
Once the membrane was in place, the team added human induced pluripotent stem cell derivatives into the chip. They observed that these cells were able to send signals across the ultrathin membrane, which helped the cells differentiate into glomerular cells, podocytes and vascular endothelial cells.
The platform also triggered the development of endothelial fenestrations in the growing tissue, which are holes that allow for the passage of fluid between the cellular layers.
By the end of the test, these different kidney cell types had assembled into a glomerular capillary wall and could efficiently filter molecules by size.
"The new microfluidic chip system's ability to simulate in vivo-like tissue-tissue interfaces and induce the formation of specialized cells, such as fenestrated endothelium and mature glomerular podocytes from stem cells, holds significant potential for advancing our understanding of human organ development, disease progression, and therapeutic development," said Musah.
As they continue to optimize their model, Musah and colleagues are hoping to use this technology to better understand the mechanisms behind kidney disease. Despite affecting more than 15% of American adults, researchers lack effective models for the disease. Patients are also often not diagnosed until the kidneys have been substantially damaged, and they are often required to undergo dialysis or receive a kidney transplant.
"Using this platform to develop kidney disease models could help us discover new biomarkers of the disease," said Mou. "This could also be used to help us screen for drug candidates for several kidney disease models. The possibilities are very exciting."
"This technology has implications for all organ-on-a-chip models," said Musah. "Our tissues are made up of membranes and interfaces, so you can imagine using this membrane to improve models of other organs, like the brain, liver, and lungs, or other disease states. That's where the power of our platform really lies."
More information: Xingrui Mou et al, An ultrathin membrane mediates tissue-specific morphogenesis and barrier function in a human kidney chip, Science Advances (2024). DOI: 10.1126/sciadv.adn2689
Journal information: Science Advances
Provided by Duke University