This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Novel design approach achieves nonlinear photochromism using easy-to-synthesize rhodamine spirolactam derivatives
Photochromic compounds, which change their color when exposed to light, have been widely used as photo switches to control different properties of materials. Nonlinear photochromic compounds, characterized by a nonlinear response to the intensity of incident light, have attracted special attention among researchers as the nonlinearity leads to enhanced contrast and improved spatial resolution in photochromic reactions.
It also allows for multiple photochromic properties in a single molecule with a single light source. These qualities have made them valuable in nonlinear optical and holographic elements, super-resolution microscopy, and biomedical applications.
The simplest way to achieve nonlinear photochromism in materials is through simultaneous two-photon absorption, but this requires extremely high-intensity light. For nonlinear photochromic reactions using low-intensity light, stepwise two-photon processes are required, but these are difficult to design because they rely on extremely short-lived molecular species.
Moreover, multi-photochromic systems that exhibit stepwise photochemical reactions require complex molecular structures. Such complexities have limited the wide applications of nonlinear photochromic compounds in many fields.
Another important method for inducing nonlinear photochromism is triplet–triplet annihilation (TTA). It requires three components: a triplet sensitizer, an annihilator, and a photochromic compound, which adds significant complexity. If a single molecule can fulfill these roles, nonlinear photochromism can be achieved in simpler systems.
In a recent breakthrough, a team of researchers from Japan, led by Professor Yoichi Kobayashi from the Department of Applied Chemistry at the College of Life Sciences at Ritsumeikan University, achieved nonlinear photochromism with low-intensity light using TTA in a single molecule.
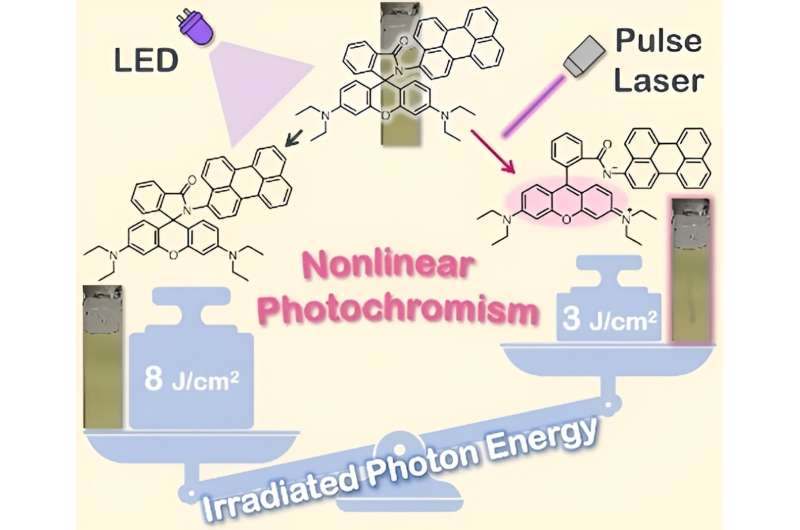
Prof. Kobayashi explains, "Rhodamine spirolactams can play the roles of both photochromic compounds and triplet sensitizers, which addresses the issue of complexity, and they can be easily synthesized from rhodamine B and its analogs. In this study, we focused on a rhodamine spirolactam derivative having a perylene group (Rh–Pe) and investigated its photochromic properties."
Their findings were published in the journal Angewandte Chemie International Edition on April 10, 2024.
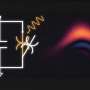
In Rh–Pe, excitation with light triggers a photochromic reaction, leading to the formation of a ring-opening structure, called open form, which results in drastic changes in its color. Upon studying its nonlinear photochromic properties, the researchers found that the color change efficiency of Rh–Pe increased with higher-intensity light.
This means that the intensity of coloration and thus the generated amount of the open form increases non-linearly with an increase in excitation intensity. For example, on excitation with 365 nm light from a light-emitting diode, Rh–Pe showed almost no color change. However, excitation with a higher-intensity 355-nm nanosecond pulse laser resulted in significant coloration, even though the light had lower energy in total.
To understand the origin of these nonlinear photochromic properties, the researchers studied the excitation mechanism of Rh–Pe. They found that when directly excited with ultraviolet (UV) and blue light, Rh–Pe transitions into a charge-transfer state, which then produces a triplet excited state. This triplet excited state then undergoes TTA, forming an intensely colored open form via an intermediate state.
This TTA accounts for the nonlinear response to light intensity since it works more efficiently with higher-intensity light. Additionally, the researchers demonstrated that Rh–Pe can also exhibit photochromism with red and green light-induced photochromism by using separate triplet sensitizers, even though it can be directly excited by UV and blue light.
"Our novel design for easily synthesized nonlinear photochromic compounds will pave the way for their diverse applications, such as high-resolution photolithography, 3D printing, and high-density optical disks," says Prof. Kobayashi.
"Our results offer a novel approach for the design of photochromic compounds and functional materials with nonlinear behavior and long-wavelength responsivity that efficiently utilize low-energy light."
Overall, the findings of the study offer new avenues for developing simpler nonlinear photochromic compounds, paving the way for wider applications.
More information: Genki Kawai et al, A Nonlinear Photochromic Reaction Based on Sensitizer‐Free Triplet–Triplet Annihilation in a Perylene‐Substituted Rhodamine Spirolactam, Angewandte Chemie International Edition (2024). DOI: 10.1002/anie.202404140
Journal information: Angewandte Chemie International Edition
Provided by Ritsumeikan University