This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
One-pot strategy to simultaneously achieve heterodehydrocoupling of hydrostannane and reduction of quinoline
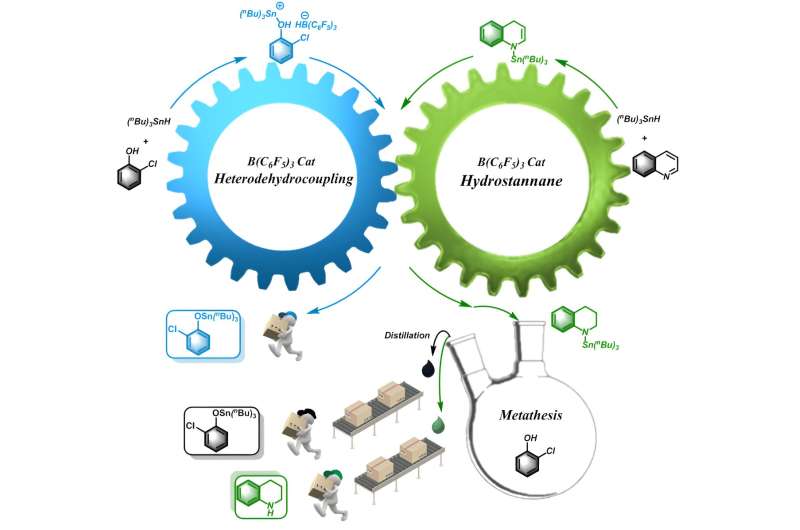
Heteroatom tin compounds (SSn, OSn, NSn, PSn) composed of heteroatoms S, O, N, P and tin atoms have attracted intense attention due to their wide applications in organic synthesis and pharmaceutical fields. The current methods for synthesis of such compounds, such as metathesis reactions, addition reactions, and free radical reactions, exhibit drawbacks including narrow substrate scope and harsh conditions. Therefore, it is important to develop efficient synthetic systems to construct heteroatom-tin bond.
Tetrahydroquinoline, as an important organic synthetic intermediate and pharmaceutical intermediate, is of significant importance in the fields of biochemistry and pharmaceutical chemistry. The commonly used method is to reduce the quinoline to tetrahydroquinoline by H2, but often requires harsh reaction conditions, such as high temperature and pressure. Therefore, the development of a highly efficient reaction system to realize the reduction of quinoline to tetrahydroquinoline under mild conditions is desirable.
In nature, there is sometimes a mutually beneficial relationship between two or more species. For example, a sea anemone is attached to the shell of a host crab and carried by the host crab, allowing it to capture foods more effectively. Meanwhile, a sea anemone uses the toxic thorn cells to protect the host crab from attacks from natural enemies.
In the Chinese Journal of Catalysis an organic synthetic strategy based on mutualism is reported by a research team led by Prof. Yuetao Zhang of Jilin University, China, in which the heterodehydrocoupling of hydrostanane and the reduction of quinolines promote each other, consuming high-energy intermediates and reducing reaction energy while expanding the substrate scope, furnishing a series of heteroatom-tin compounds and tetrahydroquinolines.
The application of mutualism in organic synthesis enabled the simultaneous accomplishment of two reactions that used to be difficult or unlikely to occur, through the mutual promotion of each other. The successful application of this mutualism concept to the organic synthesis would definitely inspire more "impossible reactions" to be tackles in the future.
More information: Tianwei Liu et al, Mutualism in organic synthetic chemistry: Simultaneous heterodehydrocoupling of hydrostannane and reduction of quinoline, Chinese Journal of Catalysis (2024). DOI: 10.1016/S1872-2067(23)64590-5
Provided by Chinese Academy of Sciences