This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Chinese scientists reveal the spinning mechanism of the silkworm
Mulberry silk is a natural protein fiber that is light, soft and fine in nature, known as the "second skin of the human body" and "Queen of fibers." China is the origin of the world's sericulture industry.
The earliest use of silk can be traced back to the Jiahu ruins about 8,500 years ago. The world-famous Silk Road opened up the first large-scale exchanges and integration of Eastern and Western civilizations in human history. China is the world's leading silk producer, both in terms of scale and output are among the world's leading.
Silk is a protein fiber with excellent mechanical properties produced by the silkworm with water-soluble silk protein as the raw material at room temperature and under normal pressure. Silk is the core of scientific research on sericulture.
For more than a century, researchers have been dedicated to resolving the spinning mechanism of silk-secreting animals such as the silkworm and spider, hoping to create artificial fibers with properties comparable to or even superior to natural silk by bionic synthesis for textile, biomedicine, military and other fields.
At present, scientists have proposed two models, "liquid crystal" and "micelle" spinning models. The former suggests that the spinning dope is a liquid crystal composed of rod-like structures formed by aggregates of globular fibroin protein, while the latter suggests that the spinning dope is a micelle formed by aggregates of amphiphilic fibroin protein.
Due to the lack of the fine structure of natural silk fibroin (NSF) in the silk gland lumen of the silkworm, the models are still highly controversial. How silkworms spin silk with excellent properties, i.e., the spinning mechanism of the silkworm, has become one of the most pressing scientific issues in sericulture science for more than 100 years.
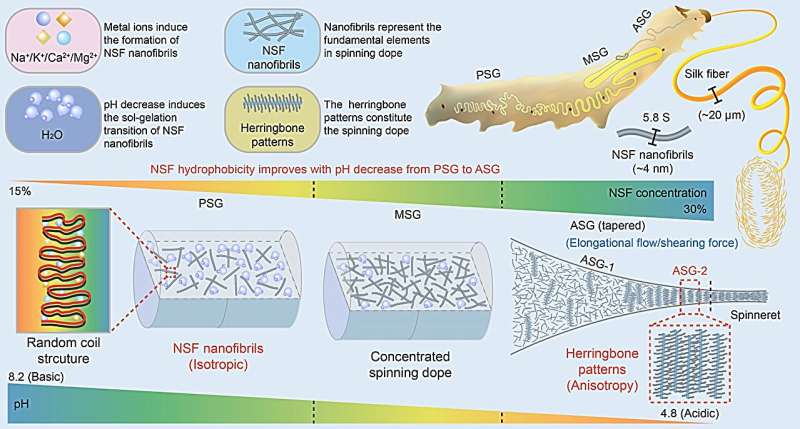
"In the silk gland lumen of the silkworm, NSF usually remains stable at concentrations of 15%–30% (w/v) without aggregation and precipitation," explained Prof. Huawei He, in the integrative science center of germplasm creation in western China (Chongqing) science city/biological science research center (SWU), who led the research.
"However, once purified, NSF quickly aggregates and forms precipitates. Therefore, most studies have used regenerated silk fibroin (RSF) rather than NSF. How to keep NSF stable in vitro, is the primary challenge in deciphering the spinning mechanism of the silkworm."
In the recent study published in Science Bulletin, they found that only amphipol and digitonin can keep NSF stable in vitro for a long term after 4 years of dissection more than 20,000 silkworm larvae and screening more than 1,000 compounds and 5,000 combinations of compounds. Then, the team systematically investigated the morphology, structure and assembly of NSF in the silk gland lumen of the silkworm.
Notably, they found that metal ions reversibly induced NSF to form flexible nanofibrils with a sedimentation coefficient of 5.8 S and a diameter of about 4 nm, instead of micelles or aggregates proposed in previous spinning models. NSF nanofibrils are mainly composed of random coils.
The successive pH decrease from the posterior silk gland (PSG) to the anterior silk gland (ASG) resulted in a gradual increase in NSF hydrophobicity, thus inducing the separation of water molecules from NSF, and the sol-gelation transition of NSF nanofibrils, which increased the concentration of NSF spinning dope thereby promoting silk fiber formation. Inspired by the discovery of graphene, they established the metal shadowing assay to determine the molecular orientation of NSF nanofibrils in situ.
Their study showed that NSF nanofibrils were randomly dispersed as isotropic nanofibrils within the lumen from PSG to ASG-1, and self-assembled into highly ordered herringbone patterns at ASG-2 near the spinneret, which are further packed together to form the anisotropic spinning dope with obvious birefringence ready for silkworm spinning.
"Our findings discovered two landmark events in the silk gland during silkworm spinning, namely, the formation of NSF nanofibrils and the self-assembly of NSF nanofibrils into highly-ordered herringbone pattern architectures programmed by pH gradient and metal ions," said Prof. He.
"Good molecular pre-alignment of NSFs contributes significantly to the toughness of silk fiber. Unlike the alignment formed by post-spinning stretching in artificial spinning, this pre-formed highly ordered molecular alignment may well explain the higher toughness of natural silk compared to that of artificial silk.
"An organized herringbone structure can effectively bear high elongation without sacrificing strength, implying that the herringbone patterns self-assembled by NSF nanofibrils may be crucial to the toughness of silk fiber. The pre-alignment of NSF nanofibrils in silkworm spinning represents a novel strategy for the fabrication of superfibers with higher strength, toughness and stiffness."
More information: Kai Song et al, Decoding silkworm spinning programmed by pH and metal ions, Science Bulletin (2023). DOI: 10.1016/j.scib.2023.12.050
Provided by Science China Press