This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Plastic recycling: Peptide with a cobalt complex oxidizes polystyrene microparticles
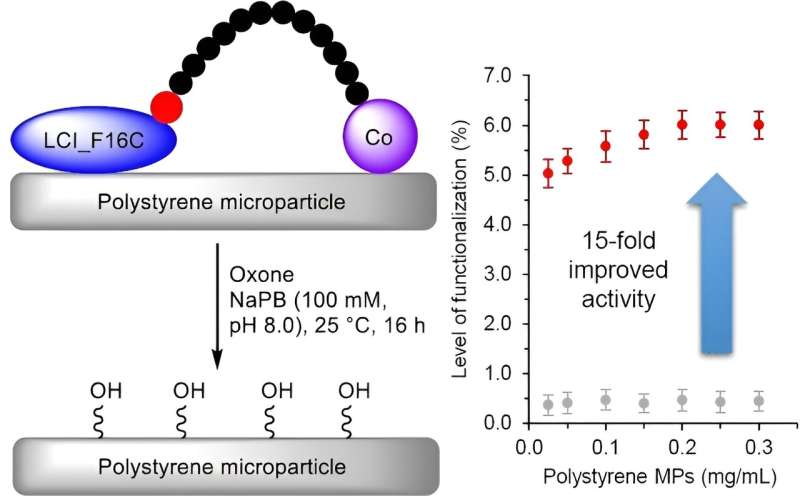
Polystyrene is a widespread plastic that is essentially not recyclable when mixed with other materials and is not biodegradable. In the journal Angewandte Chemie, a German research team has introduced a biohybrid catalyst that oxidizes polystyrene microparticles to facilitate their subsequent degradation. The catalyst consists of a specially constructed "anchor peptide" that adheres to polystyrene surfaces and a cobalt complex that oxidizes polystyrene.
Polystyrene—alone or in combination with other polymers—has many applications, from yogurt containers to instrument housings. In its foam form, mainly known under the trademarked name Styrofoam, it is, for example, used for insulation and packaging. A big disadvantage of polystyrene is its poor biodegradability, which leads to environmental pollution.
When clean and not mixed with other materials, polystyrene is recyclable, but not when it is contaminated, or combined with other materials. In municipal recycling programs, mixed polystyrene plastic waste and degradation products, such as polystyrene nano- and microparticles, are difficult to process. The problem lies in the fact that polystyrene is water-repellent and nonpolar and thus cannot react with common polar reactants.
For a simple, economical, and energy-efficient process to break down mixed polystyrene waste, the polystyrene must first be equipped with polar functional groups. A team led by Ulrich Schwaneberg and Jun Okuda at the RWTH in Aachen (Germany) has now developed a novel biohybrid catalyst to carry out this step. The catalyst is based on compounds known as anchor peptides coupled with a cobalt complex.
Anchor peptides are short peptide chains that can attach to surfaces. The team developed a special anchor peptide (LCI, Liquid Chromatography Peak I) that binds to the surface of polystyrene. One gram of this peptide is enough to coat a surface of up to 654 m2 with a monolayer within minutes by either spraying or dipping.
A catalytically active cobalt complex is attached to the anchor peptide via a short linking piece. The cobalt atom is "surrounded" by a macrocyclic ligand, a ring made of eight carbon and four nitrogen atoms (TACD, 1,4,7,10-tetraazacyclododecane). The catalyst accelerates oxidation of the C–H bonds in polystyrene to form polar OH groups (hydroxylation) by reaction with Oxone (potassium peroxymonosulfate), a common oxidizing agent.
The binding of the anchor peptides is material-specific so in this case they immobilize the catalytically active cobalt near the polystyrene surface, which accelerates the reaction. This simple, inexpensive, and energy-efficient process is scalable through dipping and spray applications and is suitable for use on an industrial scale.
Through the use of conjugated chemical catalysts, this hybrid catalyst concept employing material-specific binding by anchor peptides could allow for the material-specific breakdown of further hydrophobic polymers such as polypropylene and polyethylene that cannot be economically broken down by enzymes.
More information: Dong Wang et al, Engineered Anchor Peptide LCI with a Cobalt Cofactor Enhances Oxidation Efficiency of Polystyrene Microparticles, Angewandte Chemie International Edition (2024). DOI: 10.1002/anie.202317419
Journal information: Angewandte Chemie International Edition , Angewandte Chemie
Provided by Wiley