This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
AI-driven lab speeds catalysis research

Researchers have developed a "self-driving" lab that uses artificial intelligence (AI) and automated systems to provide in-depth analyses of catalytic reactions used in chemical research and manufacturing. The new tool, called Fast-Cat, can provide more information in five days than is possible in six months of conventional testing.
The research paper, "Autonomous reaction pareto-front mapping with a self-driving catalysis lab," appears in the journal Nature Chemical Engineering.
At issue are the yield and selectivity of chemical reactions in the presence of molecules called ligands.
Yield refers to how efficiently a chemical reaction produces a desired product from the chemicals you started with. Selectivity refers to the extent to which you can make a chemical reaction produce a specific product instead of creating multiple products. Ligands are widely used in catalysis, speeding up and controlling the selectivity of chemical reactions used in processes ranging from industrial chemistry to pharmaceutical manufacturing.
From an industry perspective, you want the highest possible yield and selectivity. Because the specific steps you take when conducting the catalytic reaction can influence both yield and sensitivity, industrial chemists spend a tremendous amount of time and effort trying to find the parameters necessary to achieve the most desirable reaction outcome.
"The problem is that conventional catalyst discovery and development techniques are time-, material- and labor-intensive," says Milad Abolhasani, corresponding author of a paper on the work and an associate professor of chemical and biomolecular engineering at North Carolina State University.
"These techniques rely largely on manual sample handling with batch reactors, as well as human intuition and experience to drive the experimental planning. In addition to the material inefficiencies, this human-dependent approach to catalyst development creates a large time gap between performing the reaction, characterizing the product, and making a decision about the next experiment. That's why we created Fast-Cat. We're now able to better understand how a specific ligand performs in five days than was previously possible in six months."
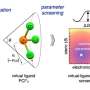
Fast-Cat is completely autonomous, using AI and automated systems to continuously run high-temperature, high-pressure, gas-liquid reactions. The autonomous technology also analyzes the output from each of these reactions to determine—with no human intervention—how different variables affect the outcome of each experiment.
Fast-Cat uses the results from all of the previous experiments it has run—both successes and failures—to inform which experiment it will run next.
"Fast-Cat's AI is constantly evolving, learning from the experiments it has already conducted," Abolhasani says.
In layman's terms, users let Fast-Cat know what ligands and precursor chemicals it has to start with, and then see how much it can learn over 60 experiments.
"We spent a lot of time fine-tuning Fast-Cat's AI model to optimize its ability to provide the broadest possible understanding of how different parameters affect the selectivity and yield of catalytic reactions using a specific ligand," Abolhasani says.
"We also spent a lot of time ensuring that Fast-Cat's findings are scalable. Fast-Cat conducts its experiments with extremely small sample sizes. But if we want its findings to be relevant for practical use, we needed to know that Fast-Cat's findings hold true for reactions conducted on the large scales that are relevant for industrial manufacturing."
For proof-of-concept testing, the researchers used Fast-Cat to characterize the catalytic performance of six ligands that are already found in the research literature.
"This technology is providing in-depth optimization of each unique ligand," says Dawn Mason, global external innovation manager at Eastman, a global specialty materials company that supported the work. "For the first time, we are able to quickly assess a wide variety of parameters and gain a truly in-depth understanding of how to influence the performance of each ligand. We more than doubled the array of possible selectivity and yield endpoints for each ligand examined. Incredibly, it only took five days to assess each one."
"There is genuine value for the chemical and pharmaceutical industries in better understanding how to influence the catalytic processes they use in manufacturing," says Jeff Carbeck, vice president of corporate innovation at Eastman. "Fast-Cat provides that understanding—and does so quickly, efficiently, and while using tiny amounts of the relevant ligands and chemical precursors. In other words, it's fast, inexpensive, and very effective."
The researchers have made the software and hardware publicly available so that Fast-Cat can be used to facilitate additional research.
"We hope other researchers can adopt this technology to accelerate catalysis discovery in academia and industry," Abolhasani says.
The paper was co-authored by Jeffrey Bennett, a postdoctoral researcher at NC State; Negin Orouji and Sina Sadeghi, who are both Ph.D. students at NC State; Muzammil Khan, a former postdoctoral researcher at NC State; and Jody Rodgers of Eastman.
More information: Autonomous Reaction Pareto-Front Mapping with a Self-driving Catalysis Lab, Nature Chemical Engineering (2024). DOI: 10.1038/s44286-024-00033-5. www.nature.com/articles/s44286-024-00033-5
Journal information: Nature Chemical Engineering
Provided by North Carolina State University