This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Scientists find a close-loop recycling process for one of the most widely used plastics
A pioneering development enables the targeted replication of the chemical structure of low-density polyethylene (LDPE), a plastic that has been difficult to imitate until now, and shows great potential for sustainable alternatives in the plastics industry.
Prof. Dr. Rhett Kempe, Chair of Inorganic Chemistry II—Catalyst Design, Sustainable Chemistry Center, at the University of Bayreuth, and his interdisciplinary research team present this material in the journal Advanced Science.
"We have introduced a new chemically recyclable, highly branched polyolefin material," explains Prof Dr. Rhett Kempe.
His team has incorporated so-called "recycling points" into the new plastic, at which the polymer can be chemically broken down into smaller fragments that are soluble in organic solvents at moderate temperatures and can therefore be recycled. The components can then be recombined, allowing them to be reused in a closed cycle.
LDPE is produced in a high-pressure process under extreme reaction conditions (at 250°C with 2,500 to 4,000 bar) via the free radical polymerization of ethylene. This energy-intensive process is crucial for the highly branched and complex chemical structure and the associated material properties.
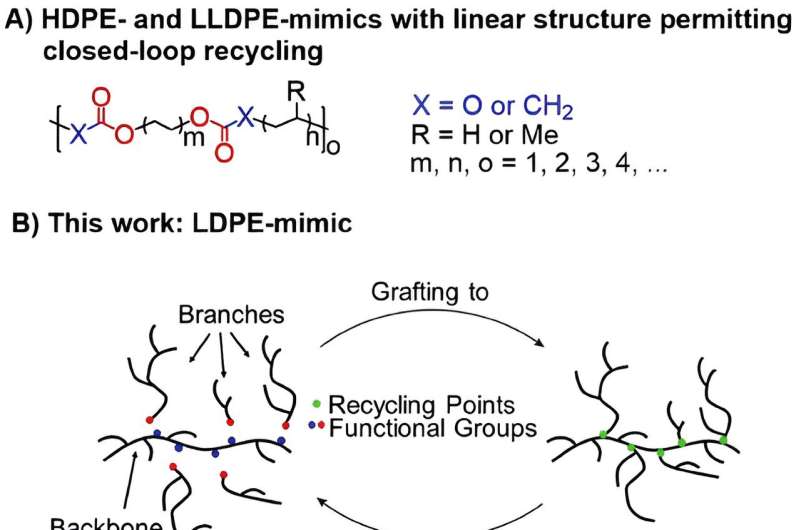
Until now, it has been very difficult to imitate this unique structure. The new material, known as LDPE-mimic, is close to commercial LDPE in its chemical structure.
"The key to success is the use of our new catalysts, which produce defined building blocks of a certain size under correspondingly mild reaction conditions, around 70° Celsius and two bar pressure. These can then be combined to form the final plastic material," says Kempe.
"The new material consists of two different macromonomers, a backbone and potential long-chain branches. The branches can be reversibly attached to the backbone and cleaved under acidic and basic conditions." A macromonomer is a compound that has the structure of a monomer (i.e. it can combine to form a larger molecule) but is already larger and has some macromolecular properties. This means that it already has a considerable size or complexity, but still has the ability to cross-link or polymerize further.
Overall, the innovations of the Bayreuth work therefore lie in the combination of production under very mild or sustainable conditions, the chemical recyclability of the plastic and the targeted imitation of the chemical structure of LDPE.
More information: Christoph Unger et al, A Closed‐Loop Recyclable Low‐Density Polyethylene, Advanced Science (2024). DOI: 10.1002/advs.202307229
Journal information: Advanced Science
Provided by Bayreuth University