This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Titanium oxide material can remove toxic dyes from wastewater
Discharged in large quantities by textile, cosmetic, ink, paper and other manufacturers, dyes carry high-toxicity and can bring potential carcinogens to wastewater. It's a major concern for wastewater treatment—but researchers in Drexel University's College of Engineering may have found a solution, using a tiny nanofilament.
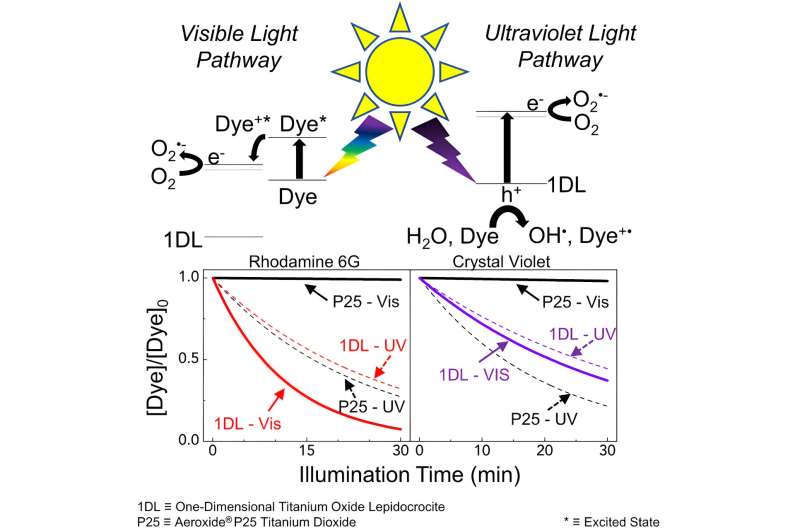
A study lead Michel Barsoum, Ph.D., Distinguished University professor in the College of Engineering, and his team, including researchers from Drexel's College of Arts and Sciences, found that a one-dimensional, lepidocrocite structured titanium oxide photocatalyst material has the ability to break down two common dye pollutants—rhodamine 6G and crystal violet—under the visible light spectrum.
The material also reduced those dye concentrations in the water by 90% and 64%, respectively, in just 30 minutes, when the starting catalyst to dye mass ratio was 1 to 1.
"This is an exciting finding because it helps to address a problem that has been a real challenge for the water treatment process," Barsoum said. "We anticipate that integrating our titanium-oxide photocatalyst into the current processes could improve its effectiveness in removing these chemicals, as well as reducing the amount of energy required to do so."
The process starts with adsorption, where the dye adheres to the surface of the nanofilament, and once illuminated undergoes photocatalysis. The dye sensitizes the nanofilaments to visible light. This process accelerates degradation, allowing the dye to break apart into harmless byproducts such as carbon dioxide and water.
The study, recently published in the journal Matter, found that the key to the dye degradation and self-sensitization process was the ability of the material to generate electron holes and something called "ROS"—hydroxyl , superoxide and singlet oxygen, radicals, as well as electron "holes."
The two dye targets are commonly appearing dye driven effluents in wastewater. Effluent, which literally means something that flows out, is different than sewage found in wastewater. Solid waste can be filtered, removed before the water is purified. Effluent is suspended in the water, making it hard to separate and remove.
Rhodamine 6G is a xanthene-derived dye primarily used in wood processing, paper dyeing, pen ink and cosmetics. Crystal violet, a triphenylmethane dye, is used to dye ink and textiles. These dyes are water soluble and any excess is discharged as effluent.
Wastewater is a major environmental concern worldwide and its existence has long-term impacts on health of humans, aquatic plants and animals. Households and industry generate nearly 380 billion cubic tons of wastewater globally each year. Only 24% of this is treated sufficiently due to challenges in treatment, including high energy consumption, the existence of residual chemicals, treatment center staffing and the insufficient processing of complex and persistent contaminants, including dyes.
The most common wastewater treatment methods, such as sedimentation, biological oxidation and chemical-physical treatment, are ineffective at removing dyes, according to the researchers, due to the dyes' complex molecular structure and water-soluble nature.
Adsorption with clay materials, activated carbon, iron oxide and natural materials such as coffee grounds, has also been used before and exhibit high cationic dye uptake, exchanging ions or forming bonds. However, these materials simply allow separation of the dye from the water—the dye still exists and is simply attached to the adsorbent materials within the wastewater.
Photocatalysts, long thought to be the key to removing dyes from water, thus far have not produced a sustainable solution. According to Barsoum, many photocatalysts typically require UV light treatment, which uses extensive energy. The impact of the new nanofilament resides in its self-sensitization behavior, which makes the nanofilament more sensitive to visible light.
"The use of visible light—light the human eye can see—like the sun or other simulated light sources, could significantly reduce the financial and energy consumption costs associated with treatment, while still being highly effective at removing dyes from wastewater, eliminating the toxic effluents," said Adam Walter, a doctoral student in Barsoum's research group, and the first author on the paper, in the Department of Materials Science and Engineering.
"This also presents an exciting opportunity for expansion into other fields like solar cells or optical devices."
The result: cleaner water without the use of additional toxins or additional energy.
To perform the study, the team used X-ray diffraction to characterize the arrangement of atoms in the nanomaterial. They further characterized the nanomaterial with scanning and transmission electron microscopy, which sends beams of electrons at the material to form an image.
To monitor dye decolorization, the team monitored the sample using ultraviolent-visible spectroscopy and quantified mineralization by chemical oxygen demand. The study details the structural and optical properties of the nanofilaments, as well as the promise of the material for wastewater treatment due to its adsorption efficiency of both dyes tested in the study.
One of the most important findings of the study was the strong evidence that the nanofilament is sensitized by the dye, representing a symbiotic relationship of additive and effluent that resulted in cleaner, less toxic water. One way of thinking about this, Walter said, is that the dye catalyzes in its own destruction.
In addition, while this study showed proof that the nanofilament could be leveraged to improve water treatment capability, it also serves as first proof that the materials can be sensitized, opening the door to other applications in solar cells and optical devices. Earlier this year, the same nanofilament was studied by the team and found to harness sunlight for hydrogen separation, which could unlock its potential in green fuel generation.
"We are just beginning to uncover the possibilities of this material," Barsoum said. "As we better understand the processes enabling its behavior, we anticipate exploring new applications where it could improve the performance of technology that the world needs to move toward a more sustainable future."
More information: , Adsorption and Self-Sensitized, Visible Light, Photodegradation of Rhodamine 6G and Crystal Violet by One-Dimensional Lepidocrocite Titanium Oxide, Matter (2023). DOI: 10.1016/j.matt.2023.09.008. www.cell.com/matter/fulltext/S2590-2385(23)00465-4
Journal information: Matter
Provided by Drexel University





















