This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Scientists decipher a central mechanism of energy production in the human body
Some two and a half thousand years ago, the Chinese general Sun Tzu wrote in the Art of War, "Know your enemy and know yourself, then you need not fear the outcome of a hundred battles." And what applied to the battlefields of ancient China also seems to apply in biomedical research.
In the case of Martin van der Laan, professor of medical biochemistry at Saarland University, and Alexander von der Malsburg, a research associate at the same institute, the enemies are hereditary defects in the protein OPA1. OPA1 plays a crucial role optimizing energy conversion in the mitochondria, which are often referred to as the "powerhouses" that drive our cells.
Given the importance of OPA1, defects in the protein can have very serious consequences. For instance, if OPA1, which stands for "Optic Atrophy 1," does not function properly, serious degenerative diseases can result. In many of these cases, it is the optic nerve that is primarily affected and patients with OPA1-related mitochondrial dysfunction often lose their sight.
But until recently studying these defective OPA1 proteins proved extremely challenging, in part because knowledge about the functionality of even healthy OPA1 was still fragmentary. Individual proteins are by their very nature far smaller than the tiny cellular compartments in which they are active and are thus not easy to observe.
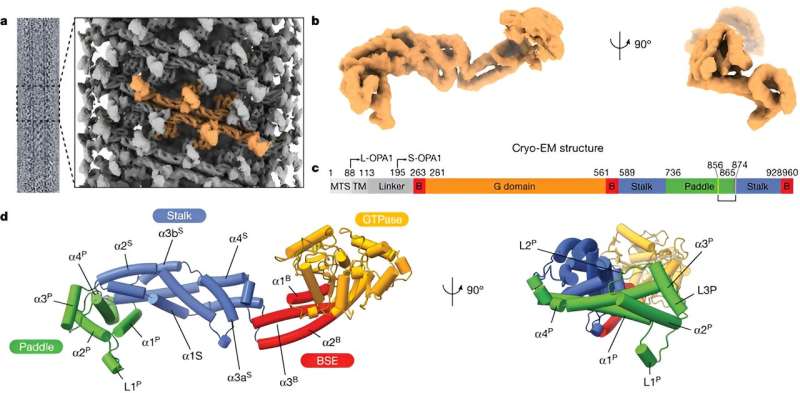
Recently, however, researchers at the University of California were able to produce the first images of OPA1 using high-resolution cryo-electron microscopy. They showed these images to Professor van der Laan and his team in Homburg, as the group enjoys an excellent global reputation in the field of mitochondrial research. Careful analysis of the new image data provided the first indications of how OPA1 might function.
The crucial breakthrough came from Alexander von der Malsburg, who succeeded in establishing the world's first cellular system for studying the function of human OPA1. Martin van der Laan praised his colleague's "very smart and elegant solution to the problem" and the insights gained are now being published in Nature.
Until now, OPA1 has been seen as a difficult protein for scientific study as it occurs in different forms and behaves in a very dynamic way. A few aspects of OPA1 functionality had been elucidated previously by studying specially prepared mouse cells created from embryonic stem cells in a complex procedure.
But there were still aspects that seemed inconsistent or contradictory and much that was completely unknown. By cleverly combining and improving a number of genetic and biochemical methods, Alexander von der Malsburg has managed to tame the human OPA1 protein and make it more readily accessible for precise scientific examination. According to van der Laan, von der Malsburg has mastered a "technically extremely challenging" task with flying colors.
The OPA1 protein strongly influences the efficiency of energy production in cellular mitochondria and thus plays a particularly important role in determining cell performance. OPA1 ensures that healthy mitochondria can fuse with each other and thus concentrate their forces, while defective mitochondria are discarded. Mitochondrial fusion is initiated when the OPA1 protein attaches to the inner membrane of the mitochondria, opening the membrane in a controlled and localized manner.
If neighboring mitochondria are modified in this way, they can fuse with each other and thus optimize mitochondrial functionality in the cell. However, if mitochondrial fusion is inhibited due, for example, to a genetic deficiency that results in the production of defective OPA1, this can seriously impact mitochondrial energy metabolism and, with advancing age, brings with it the risk of severe degenerative diseases.
"There are dozens of different variants of defective OPA1," explained Martin van der Laan. Precise knowledge of how the OPA1 protein acts and the ability to conduct experimental simulations of malfunctioning OPA1 could potentially help many patients in future.
So just how does OPA1 function? "Working with our American partners, we found that OPA1 initially attaches to the inner membrane like a foot with a 'claw-like' structure and then lifts the 'heel' of the foot," said Alexander von der Malsburg. This mechanism pulls a chunk out of the membrane envelope in a manner not unlike the way a lever corkscrew lifts the cork out of the neck of a bottle of wine.
The proof that this mechanism is crucial for OPA1 functionality was ultimately achieved by manipulating the gene that contains the blueprint for producing the OPA1 protein. The research team were able to smuggle modified genetic blueprints into healthy human cells so that they began to create defective variants instead of healthy OPA1. "After a while, we began to observe that the cells' energy supply mechanism was impaired and that mitochondrial fusion was malfunctioning," explained Alexander von der Malsburg.
He described what they found under the microscope: "It was apparent that the 'claw-like' structure was completely missing in the genetically manipulated version." The manipulated OPA1 protein was now no longer able to open the membrane, essentially preventing mitochondrial fusion—the protein had effectively switched cellular roles: from crucial ally to dangerous adversary.
"This is a fundamental mechanism that affects numerous variants of defective OPA1," said Professor Martin van der Laan. "And we now have the means to study all these variants individually." These new research findings could help pave the way to customized therapeutic solutions for patients who become ill due to loss of function of OPA1.
It is already possible to carry out genetic examinations of patients to determine which of the many known OPA1 defects they have. Van der Laan summarized the scope of the work as follows, "With this new, much improved understanding of the OPA1 protein, we're hopeful that in future patients will be able to receive treatments that are targeted at the specific underlying protein defect."
By uncovering the mechanism by which defects in the OPA1 protein lead to mitochondrial dysfunction, Alexander von der Malsburg and Martin van der Laan have complied with the first part of Sun Tzu's famous dictum "Know your enemy." And after collaborating so successfully, it's probably fair to say that they also know themselves just as well. Having essentially fulfilled both parts of Sun Tzu's maxim, the researchers look well set for the next hundred research battles with dysfunctional OPA1 variants.
More information: Alexander von der Malsburg et al, Structural mechanism of mitochondrial membrane remodelling by human OPA1, Nature (2023). DOI: 10.1038/s41586-023-06441-6
Journal information: Nature
Provided by Saarland University