This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Electron-rich metals make ceramics tough to crack

Researchers have discovered a way to make ceramics tougher and more resistant to cracking. By building these materials using a blend of metal atoms possessing more electrons in their outer shell, a team led by engineers at the University of California San Diego has unlocked the potential to enable ceramics to handle higher levels of force and stress than before.
Ceramics offer many advantages due to their remarkable properties, including their ability to withstand extremely high temperatures, resist corrosion and surface wear, and maintain lightweight profiles. These properties make them suitable for a variety of applications such as aerospace components and protective coatings for engines and cutting tools. However, their weakness has always been their brittleness. They break easily under stress.
But now, researchers have found a solution that could make ceramics harder to break. They published their work recently in Science Advances.
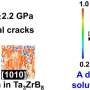
The study, led by UC San Diego nanoengineering professor Kenneth Vecchio, centers on a class of ceramics known as high-entropy carbides. These materials have highly disordered atomic structures, composed of carbon atoms bonded with multiple metal elements from the fourth, fifth and sixth columns of the periodic table.
These metals include titanium, niobium and tungsten, for example. The researchers found that the key to enhancing ceramic toughness lay in the use of metals from the fifth and sixth columns of the periodic table, due to their higher number of valence electrons.
Valence electrons—those residing in an atom's outermost shell and engaging in bonding with other atoms—proved to be a pivotal factor. By using metals with a higher valence electron count, the researchers successfully improved the material's resistance to cracking when subjected to mechanical load and stress.
"Those extra electrons are important because they effectively make the ceramic material more ductile, meaning it can undergo more deformation before breaking, similar to a metal," said Vecchio.
To better understand this effect, Vecchio's group collaborated with Davide Sangiovanni, a professor of theoretical physics at Linköping University, Sweden. Sangiovanni performed the computational simulations, and Vecchio's team experimentally fabricated and tested the materials.
The team investigated high-entropy carbides featuring various combinations of five metal elements. Each combination yielded a different concentration of valence electrons within the material.
They identified two high-entropy carbides that exhibited exceptional resistance to cracking under load or stress, thanks to their high valence electron concentrations. One was composed of the metals vanadium, niobium, tantalum, molybdenum and tungsten. The other variant substituted niobium with chromium in the mix.
Under mechanical load or stress, these materials were able to deform or stretch, respectively, resembling the behavior of metals rather than the typical brittle response of ceramics. As these materials were punctured or pulled apart, bonds began to break, forming atom-sized openings.
The additional valence electrons around the metal atoms then reorganized to bridge these openings, forming new bonds between neighboring metal atoms. This mechanism preserved the material's structure around the openings, effectively inhibiting them from growing bigger and forming cracks.
"We discovered that there's this underlying transformation happening at the nanoscale where the bonds are being rearranged to hold the material together," said study co-author Kevin Kaufmann, a UC San Diego nanoengineering Ph.D. alumnus from Vecchio's lab. "Instead of just cleaving right across the fracture surface, the material slowly frays like a rope would when it is being pulled. In this way, the material can accommodate this deformation that's occurring and not fail in a brittle manner."
The challenge now lies in scaling up the production of these tough ceramics for commercial applications. That could help transform technologies that rely on high-performance ceramic materials, from aerospace components to biomedical implants.
The newfound toughness of these ceramics also paves the way for their use in extreme applications, such as leading edges for hypersonic vehicles. Tougher ceramics could serve as frontline defense for these vehicles, shielding vital components from getting impacted by debris and enabling the vehicles to better survive supersonic flights, explained Vecchio.
"By addressing a longstanding limitation of ceramics, we can greatly expand their use and create next-generation materials that hold the potential to revolutionize our society," said Vecchio.
More information: Davide G. Sangiovanni et al, Valence electron concentration as key parameter to control the fracture resistance of refractory high-entropy carbides, Science Advances (2023). DOI: 10.1126/sciadv.adi2960
Journal information: Science Advances
Provided by University of California - San Diego