This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Unraveling metabolic processes spanning from subcellular organelles to complex tissues
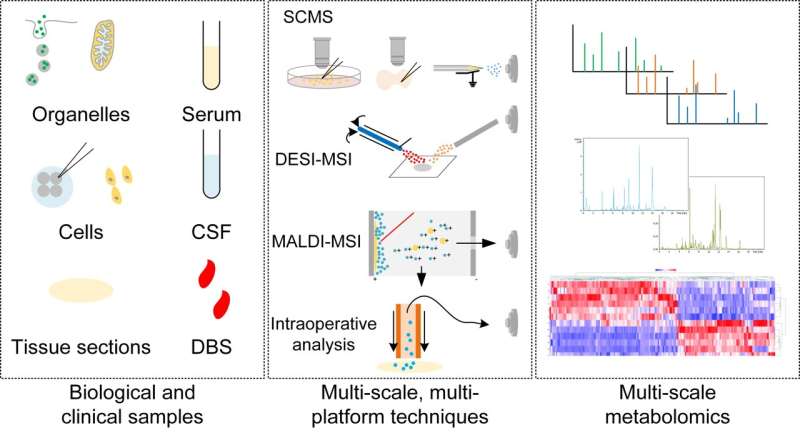
Metabolism serves as a direct reflection of biological functions and pathological conditions. Throughout the years, biologists and doctors have been delving into the intricate metabolic mechanisms underlying physiological processes and disease states. A plethora of techniques, including LC-MS, GC-MS, and CE-MS, have significantly expanded our comprehension of the pivotal role metabolism plays in both health and disease.
Nevertheless, profiling the metabolome at various scales ranging from organelles to cells and tissues remains an ongoing challenge. The conventional use of LC-MS, GC-MS, and CE-MS for analyzing homogenates of biological and clinical samples has limited our ability to uncover cellular heterogeneity within tissues.
Consequently, the metabolome of individual cells or the in situ distribution of metabolites within tissues still elude us. Fortunately, novel modifications and advancements built upon traditional techniques have greatly accelerated progress in metabolomics research while enhancing its application in both biological studies and clinical practice.
The remarkable breakthroughs, exemplified by the revolutionary single-cell and single-organelle metabolomics, provide comprehensive insights into the intricate metabolic mechanisms of individual cells and organelles. The advancements in single-cell transcriptomics and sequencing have significantly propelled the exploration of cellular heterogeneity, captivating the scientific community's attention towards profiling samples such as singular cells and organelles.
These remarkable strides enable a holistic investigation into the diverse nature of metabolism and its pivotal role in numerous biological processes, including unraveling the metabolic heterogeneity within aging lysosomes and unveiling novel pathways of metabolism.
Visualization of metabolism in situ is an additional task undertaken by biologists and clinical researchers. The alteration of metabolites in specific biological processes and diseases plays a pivotal role in comprehending the underlying metabolic mechanisms. The advancements in mass spectrometry imaging facilitate on-site metabolic profiling of tissue sections and individual cells with exceptional precision, thereby enabling the direct dissection of cellular and tissue metabolism.
These techniques have propelled significant progress in cellular biology, including the determination of differentiation fates based on cellular metabolism. Spatial reconstruction techniques offer a direct visualization of the distribution and alterations within the three-dimensional space of the metabolome. Various modifications to mass imaging have further enhanced analytical techniques for clinical applications, encompassing intraoperative metabolomics, pathological analysis, and tumor margin determination.
Conventional metabolomic techniques, such as LC-MS and GC-MS, find application in the realms of clinical therapeutic drug monitoring (TDM), newborn screening (NBS), and biomarker discovery. The modification and advancement of these techniques facilitate enhanced solutions in health care and therapeutics.
Collectively, technical advancements in metabolomics significantly contribute to the analysis of biological and clinical samples at various levels, offering a comprehensive perspective on metabolism in both health and disease. The implementation of innovative metabolomic techniques has resulted in remarkable breakthroughs in biological and clinical research, promising even greater benefits for patients in the foreseeable future.
The study is published in the journal Science Bulletin.
More information: Ziyi Wang et al, Advances in mass spectrometry-based multi-scale metabolomic methodologies and their applications in biological and clinical investigations, Science Bulletin (2023). DOI: 10.1016/j.scib.2023.08.047
Provided by Science China Press