This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Research team develops novel heat-sensitive ferritin mutant to efficiently load chemotherapy drugs
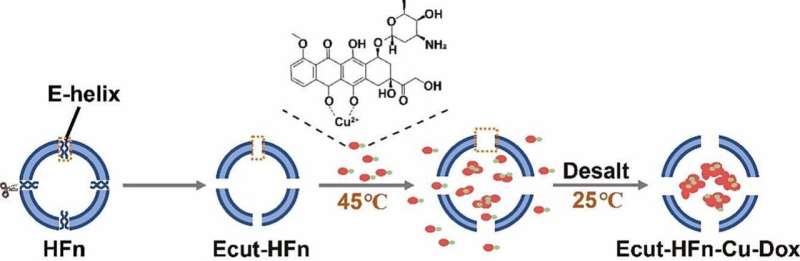
A research team led by Prof. Wang Junfeng from the Hefei Institutes of Physical Science (HFIPS), Chinese Academy of Sciences (CAS), has developed a novel heat-sensitive ferritin mutant and realized easy and efficient loading of the chemotherapy drug doxorubicin.
The findings were published in International Journal of Biological Macromolecules on Sept. 18.
Human ferritin forms a stable nanocage by itself. Its nanocage shape and natural targeting capabilities have been exploited for loading medicinal compounds, imaging agents, nucleic acids, etc. It has also been used to diagnose and treat different diseases.
However, current drug loading techniques for ferritin are complex and require strict reaction conditions including high temperatures and strong acids/bases. Ferritin-based nanomedicine has limited drug loading and protein recovery rates, limiting its clinical use.
In this study, through an in-depth analysis of the eight hydrophilic triphase channels and six hydrophobic tetraphase channels of ferritin surface, the research team predicted the correlation between the four-phase channel structure domain of ferritin and its heat sensitivity.
Based on this discovery, they developed Ecut-HFn, a unique and highly heat-sensitive ferritin mutant, and achieved one-step drug loading at low temperatures (45°C) with the help of copper ions.
"There are various advantages using this new ferritin-based medication loading strategy," said Ma Kun, member of the team.
On one hand, the entire encapsulation process had no requirement for acid/base reagents or high-temperature treatment, making it eco-friendly. It was suitable for loading biologically active compounds that were sensitive to acid/base and high-temperature conditions.
On the other hand, the shell-like structure of ferritin remained intact throughout the process, with almost no protein disassembly involved. This enabled high protein recovery rates and preserved the natural tumor-targeting ability of ferritin.
"Furthermore, there was a significant improvement in drug loading efficiency," said Ma.
More information: Haining Xia et al, Heat sensitive E-helix cut ferritin nanocages for facile and high-efficiency loading of doxorubicin, International Journal of Biological Macromolecules (2023). DOI: 10.1016/j.ijbiomac.2023.126973
Provided by Chinese Academy of Sciences