This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
The first organic oscillator that makes catalysis swing
Oscillating chemical systems are present at nearly every popular chemistry exhibition—especially the ones that display striking color changes. But so far there are very few practical uses for these types of reactions beyond timekeeping. In nature, on the other hand, many important life processes such as cell division and circadian rhythms involve oscillations.
Scientists at the University of Groningen have now developed an oscillating system that contains a catalyst, and exhibits periodic catalytic activity: This synthetic chemical oscillator can do more than just keep time. A description of this complex behavior created from simple molecules was published in Nature on 6 September.
The conventional way to perform complex chemical reactions is a step-by-step process of single reactions, with purifications between the steps. The system devised in the lab of University of Groningen Professor of Homogeneous Catalysis Syuzanna Harutyunyan takes an entirely different approach, more similar to what happens in living cells. Different components are mixed together in a flow cell where all reactions take place at the same time.
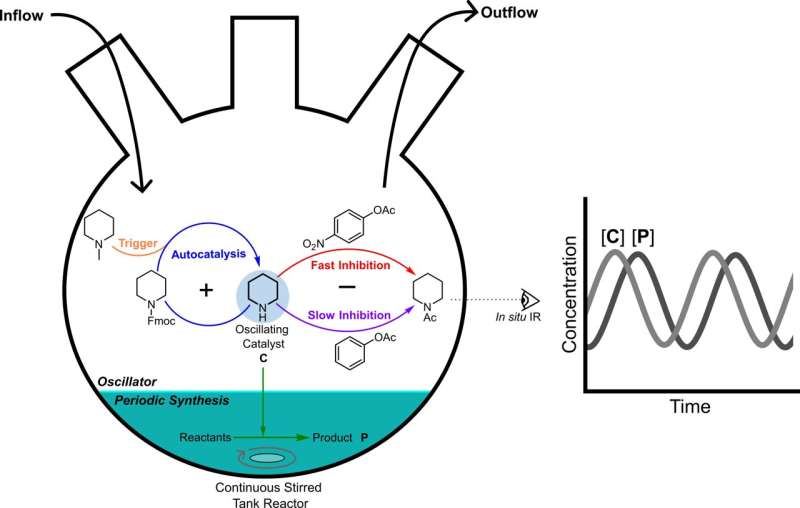
"By combining positive and negative feedback loops, we made all reactions work together to produce an oscillating system," explains Matthijs ter Harmsel, a former Ph.D. student in the Harutyunyan group. The inflow of fresh chemicals is regulated in such a way that the system remains out of equilibrium.
Trigger molecule
Ter Harmsel designed a system consisting of five different chemical reactions all occurring in the same flask where each chemical component has its unique purpose. At its core is piperidine, combined with a fluorenyl methoxycarbonyl protecting group (Fmoc). This base-labile protecting group is widely used in organic synthesis. A trigger molecule removes the protecting group and the resulting piperidine acts as a catalyst for its own formation. Two different acetates act as inhibitors, one fast, and one slow. The slow inhibitor irreversibly removes the piperidine from the mixture.
The entire process takes place inside a continuously stirred tank reactor with an inflow of reagents and an outflow of reaction products. "By combining positive and negative feedback loops, we produced a pulse, the single component of an oscillating system," explains Ter Harmsel. "Next, I used a model that contains the speed for all chemical reactions inside the reactor, which allowed me to determine at which concentrations the system would oscillate."

Two years to create the system
With some help from Oliver Maguire from the Huck lab at the Radboud University Nijmegen, the right conditions for the oscillating system were found. Together with Ter Harmsel, Maguire successfully implemented the oscillating reaction network in a flow reactor, allowing the system to reach the oscillatory phase.
As the key oscillating species is a small molecule organocatalyst, the researchers reasoned that by having it accelerate separate chemical reactions, they could be made to oscillate as well. This, in turn, would lead to enhanced chemical selectivity in coupled systems, favoring one reaction over the other in a mixture of reactions.
As a result of the oscillations, the amount of the chemicals, including piperidine organocatalyst, increases and decreases regularly.
"It took us two years to create the system, which contains a network of thirteen different components," says Syuzanna Harutyunyan. "Once we had a first pulse in the formation of our organocatalyst, we knew we could make it oscillate."
Researcher Wilhelm Huck observes, "This was a very exciting multidisciplinary collaboration, where synthetic chemistry, catalysis, physical organic chemistry, and theory, all contributed to make a chemical system 'tick.'"
Polymer brushes
The new oscillator is not designed for nice color changes, but to do real work, such as autonomous time-controlled periodic synthesis. Biologists wouldn't be surprised by this, as many biological systems show oscillations with coupled processes. Ter Harmsel says, "However, to organic chemists or those working in catalysis, it is a strange concept." Ter Harmsel is interested in systems chemistry, an area that is more like biology.
The oscillating system might also have practical applications, and could for example be used to synthesize polymers, explains Sofiya Runikhina, a postdoc in the group: "We are working on that right now. We want to add beads to the reactor on which polymers can grow. At each oscillation, monomers are added to the growing chain. This is a very neat way to grow dense polymer brushes."
Possible applications
Other applications, Ter Harmsel adds, could be in drug delivery. "Lots of drugs have structures resembling piperidine. Some hormones might act as triggers, like the ones we use to start the oscillations."
Additionally, the system could be used as a filter, in which unwanted products react fast and disappear, while the desired molecule does not have time to react during the short bursts when the catalyst is present and will therefore be present in the efflux of the reactor.
"We have demonstrated that it is possible to create a complex system that combines oscillations and catalysis with very simple molecules, that are present in almost any organic chemistry lab," concludes Harutyunyan. This opens up all kinds of possibilities. "A system like this has never been described, so we anticipate many new developments. There may be potential applications that we haven't even thought about yet."
More information: Matthijs ter Harmsel et al, A catalytically active oscillator made from small organic molecules, Nature (2023). DOI: 10.1038/s41586-023-06310-2. www.nature.com/articles/s41586-023-06310-2
Journal information: Nature
Provided by University of Groningen