This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Electron transfer kineties of CdS/Zn(impim) dots-on-rods designed for efficient visible-light reduced C-X bond
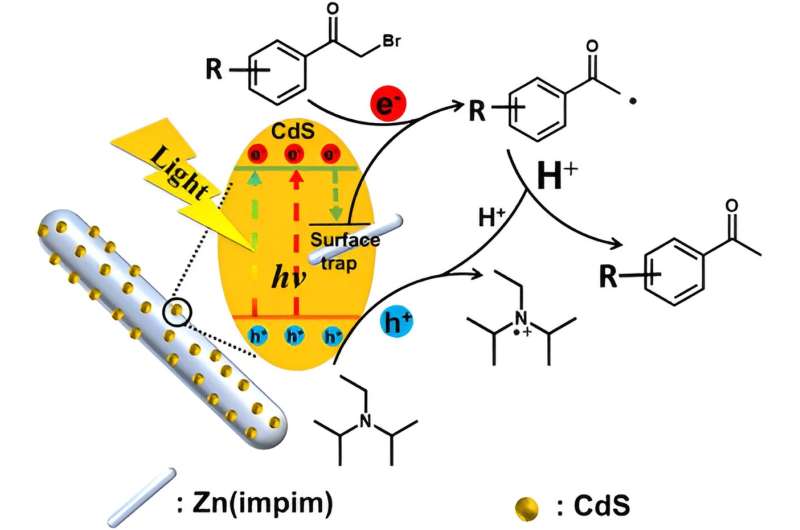
Using solar energy to drive the cleavage of C-X bonds in halogenated organic compounds to form C-H bonds can not only control environmental pollution but also achieve important organic conversion reactions.
In a study published in ACS Applied Materials & Interfaces, a research group led by Prof. Cao Rong from Fujian Institute of Research on the Structure of Matter of the Chinese Academy of Sciences reported a CdS/Zn(impim) coupled system in which the electron transfer kinetics of photocatalytic reduction of C-X bond for CdS/Zn(impim) composites has been explored.
The researchers synthesized CdS/Zn(impim) (MOF) dots-on-rods composite photocatalyst under mild conditions, and characterized the structure and morphology by powder X-ray diffraction (PXRD), scanning electron microscope (SEM) and transmission electron microscope (TEM).
They found that CdS nanoparticles are uniformly loaded on the surface of rod-shaped Zn(impim) MOF, promoting the separation of electron-hole pairs.
To prove the direction of electron transfer from CdS to Zn(impim), the researchers measured the surface work functions by the Kelvin probe force microscope (KPFM). The fluorescence lifetime of CdS/Zn(impim) was significantly reduced compared to CdS. The related electron transfers kinetic was specified in detail by fluorescence spectrum and femtosecond transient absorption spectroscopy (fs-TAS).
In order to demonstrate the photocatalytic performance of CdS/Zn(impim), the researchers selected α-bromoacetophenone, bromobenzonitrile, and their derivatives as model reactions.
They revealed that the photocatalytic dehalogenation activity of CdS/Zn(impim) composite is much higher than that of CdS or Zn(impim) alone. The catalyst enjoyed good stability, and the morphology and structure of the catalyst did not change obviously after five consecutive cycles.
In the dehalogenation reaction of α-bromoacetophenone, the researchers detected the intermediate of acetophenone radical by electron paramagnetic resonance (EPR), proving the intermediate process of the reaction.
Under visible light irradiation, the electrons in CdS are excited and transferred to Zn(impim), and holes are retained in CdS, resulting in charge separation, thereby slowing down the charge recombination process and ultimately improving the efficiency of photocatalytic dehalogenation.
This study provides a further understanding of the electron transfer mechanism in semiconductor/MOF composites and the process of photocatalytic halide dehalogenation, and a feasible idea to design higher-performance photocatalytic materials.
More information: Yanan Feng et al, Rational Design of Electron Transfer Kineties of CdS/Zn(impim) Dots-on-Rods for Efficient Visible-Light Reduced C-X Bond, ACS Applied Materials & Interfaces (2023). DOI: 10.1021/acsami.3c07110
Journal information: ACS Applied Materials and Interfaces
Provided by Chinese Academy of Sciences