This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Disrupting a core metabolic process in T cells may improve their therapeutic efficacy
In exploring an aspect of how killer T cells generate the raw materials required for their proliferation, a Ludwig Cancer Research study has uncovered an unexpected link between the immune cells' metabolism, regulation of gene expression, persistence and functional efficacy that could be exploited using existing drugs to improve cancer immunotherapy.
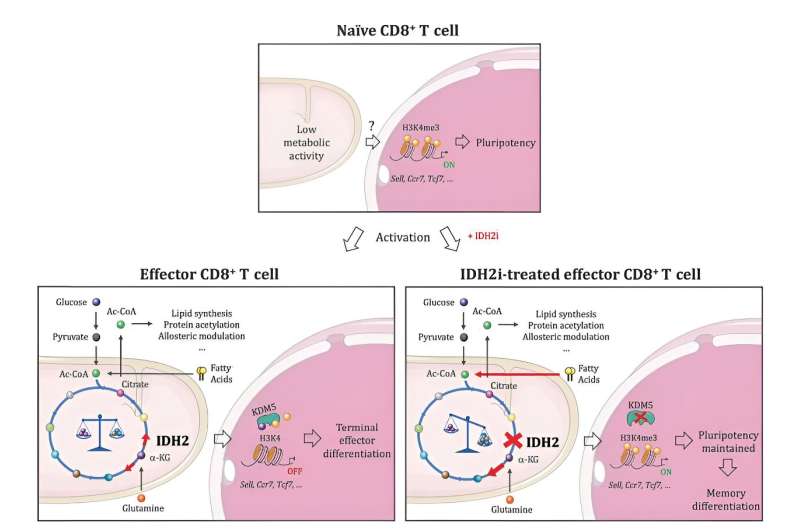
Researchers led by Ludwig Lausanne's Alison Jaccard and Ping-Chih Ho along with their University of Lausanne colleagues Mathias Wenes and Pedro Romero were exploring how proliferating T cells in the low-oxygen environment of tumors make citrate, a molecule essential to manufacturing membranes, which are required in large quantities to make new cells.
The question was whether the killer (CD8+) T cells—which destroy sick and infected cells—use a trick employed by multiplying cancer cells to ensure a steady supply of the critical molecule in similarly hypoxic conditions: shunting the amino acid glutamine through a series of chemical reactions, including one known as reductive carboxylation, to make citrate.
"We show in this study that CD8+ T cells indeed engage this metabolic pathway, and that an extensively characterized metabolic enzyme known as isocitrate dehydrogenase plays a central role in the process," said Ho.
"But what really surprised us—and could be significant for cancer immunotherapy—is that genetically or pharmacologically disrupting the enzyme did not hamper the proliferation or function of CD8+ T cells, as we expected it would. Rather, it led to the transformation of their progeny into memory T cells that are more vigorous and live far longer than their predecessors."
The researchers further report in the current issue of Nature that both mouse and human chimeric antigen receptor (CAR) T cells grown in the presence of drugs that inhibit isocitrate dehydrogenase (IDH2) take on all the key properties of memory T cells. They also demonstrate that these CAR-T cells—which are engineered to target specific markers on cancer cells—show enhanced anti-tumor activity in mouse models of melanoma, leukemia and multiple myeloma.
"When CD8+ T cells are prepared for treatments involving adoptive cell transfer, they tend to be relatively exhausted, meaning they have a limited ability to proliferate and die pretty quickly after activation," said Jaccard. "This results in their inefficient engraftment and cancer recurrence. But memory T cells persist and can proliferate over and over again when activated by their targets, making them much better instruments of CAR-T and other adoptive cell therapies."
Analysis of how disrupting IDH2 activity affected the T cells revealed a link between an altered profile of metabolites in them and epigenetic regulation of their gene expression, in which the chemical tagging of DNA and its protein packaging dynamically alters chromosome structure to determine the availability of genes for reading.
"Our studies showed that when IDH2 is inhibited by drugs, the cell engages alternative metabolic pathways to compensate," said Jaccard. "That naturally alters the types and amounts of metabolites generated in the cell, and some of the metabolites affected by these changes are involved in regulating epigenetic enzymes. This is what drives the transformation of CD8+ T cells into memory cells."
Specifically, IDH2 inhibition affects a core metabolic process known as the TCA cycle, forcing the T cells to activate compensatory metabolic pathways. This alters the profile of metabolites in the cells, boosting the levels of molecules that inhibit an epigenetic enzyme known as KDM5 and so changing the deposition of a key epigenetic "mark" on their chromosomes.
As a result, the chromosomes open up in a way that gives the cells' gene expression machinery access to genes that define memory T cells, triggering their transformation. In the absence of IDH2 inhibition, those genes are kept under wraps, bolstering the terminally exhausted CD8+ T cell identity.
Aside from their practical applications, these findings indicate that reductive carboxylation is not required for the proliferation of CD8+ T cells, as it appears to be for rapidly growing cancer cells. Rather, the metabolic process switches on gene expression programs that lock the immune cells into a terminal effector state—one in which they retain functional capability but their lifespans and proliferative capacity are curtailed.
Ho says his team now plans to examine how best to sabotage reductive carboxylation to prepare better cells for CAR-T and other adoptive T cell therapies, comparing pharmacologic inhibition with gene editing strategies to that end. He and his colleagues will also explore whether cancer cells adapt to hijack the metabolism and epigenetic regulation of T cells to push them into a less persistent, terminal effector state.
More information: Alison Jaccard et al, Reductive carboxylation epigenetically instructs T cell differentiation, Nature (2023). DOI: 10.1038/s41586-023-06546-y
Journal information: Nature
Provided by Ludwig Institute for Cancer Research




















