This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
proofread
Synthesizing a 2D copper-based complex and expanding it into a 3D structure to explore its applications
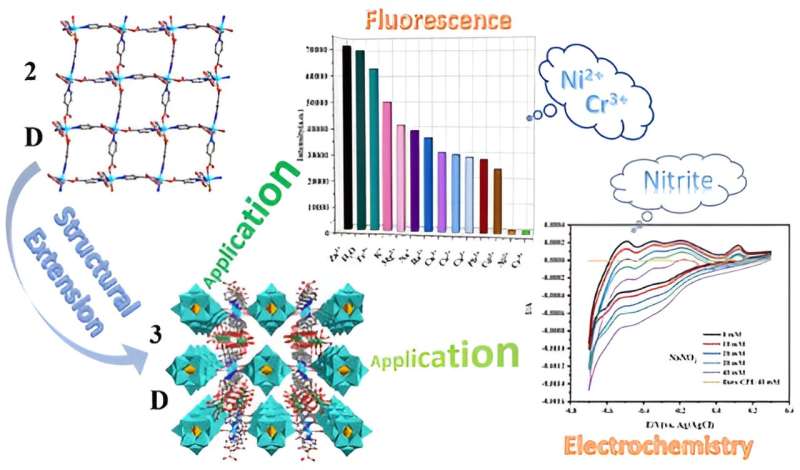
A research group has synthesized a 2D copper-based complex and expanded it into a 3D structure by adding H4SiW12O40 and rare earth metal. Through this synthesis method the team obtained three isostructural 3d−4f metal-incorporated POMs.
The team then explored the application of these complexes in the fields of fluorescence and electrochemistry. They discovered that with their expanded structure, these complexes could be used as a fluorescence sensor to detect nickel cations (Ni2+), chromium (Cr3+), and nitrite (NO2-) and an electrochemical sensor to detect nitrite. This work holds potential applications in environmental monitoring.
Their work is published in the journal Polyoxometalates.
Polyoxometalates (POMs) have a broad variety of structures and functions that makes them one of the most versatile classes of inorganic molecular materials. They are formed by bridging oxygen atoms with transition metals in their oxidation states. Their potential applications are varied, including material science, catalysis, medicine, environmental protection, and hydrogen production.
However, because POMs dissolve easily in acidic or neutral solutions, their use in various applications is limited. One possible strategy to overcome this limitation is to combine POMs with metal ions to form metal complexes.
So the team synthesized the copper-based complexes from a 2D structure to a 3D structure by adding silicotungstic acid (H4SiW12O40) and rare earth metals. They employed a hydrothermal method that involved heating the crystals for an extended period and then cooling them, and also used a ligand containing nitrogen and carboxylic acid as a binder to extend the complex from 2D to 3D.
By adding the silicotungstic acid and rare earth metal (Ln3+), they successfully produced three 3d-4f bimetallic POMs complexes. In general, 3d-4f-metal complexes are coordination compounds consisting of transition metal ions and rare earth metals called lanthanides.
Next they characterized the purity, thermal stability, and optical and electrochemical properties of the three POM complexes they obtained. They used techniques including X-ray diffraction, infrared spectrum analysis, thermogravimetric analysis, and ultraviolet-visible absorption spectra analysis to study the complexes.
They chose one of the compounds (Cu-Sm-CP) to study as a sensor. They used the compound as a fluorescence probe to identify metal cations. The team investigated the compound's ability to detect different metal ions in water at room temperature. They chose 13 metal ions for detection. They also used the compound for electrochemical sensing for nitrate.
Nitrate is a highly toxic substance that can easily form carcinogens in the human body and even leads to death. The team's results showed them that Cu-Sm-CP has a potential application in the field of electrocatalysis and can be used as an electrochemical sensor material for nitrite detection.
"We synthesized three isostructural 3d-4f POMs and selected Cu-Sm-CP among them as fluorescence and electrochemical dual-function sensors to detect Cr3+ and Ni2+ using fluorescence sensing, as well as nitrite through electrochemical sensing," said Wei Yao, with the University of Science and Technology Liaoning. In recent years, the detection of trace metal ions has attracted more and more attention because of the extremely toxic and acute poisoning caused by metal ions.
The team discovered that the Cu-Sm-CP exhibits remarkable fluorescence sensing capabilities, enabling them to identify Cr3+ and Ni2+ in aqueous solutions with exceptional recognition and anti-interference properties. "Finally, Cu-Sm-CP, as an electrochemical sensor, demonstrates excellent electrocatalytic performance for nitrite oxidation," said Yao.
The team's work not only provides a simple and feasible method for preparing 3d−4f metal-incorporated POMs complexes, but also provides effective materials for fluorescence sensing and electrochemical sensing.
Looking ahead to future research, the team hopes to expand their testing in real-world environmental conditions. "The next step is to detect the analyte in the actual environment, and finally complete the detection of harmful ions in living water, which has potential application value in environmental monitoring," said Yao.
More information: Wei Liu et al, Structural extension of 2D complexes to 3D complexes and their applications, Polyoxometalates (2023). DOI: 10.26599/POM.2023.9140032
Provided by Tsinghua University Press