This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Scientists discover how parasites of viruses drive superbug evolution
In a study published in Cell, scientists from the National University of Singapore (NUS) and Imperial College London have discovered a new way by which bacteria transmit their genes, enabling them to evolve much faster than previously understood.
Led by Assistant Professor John Chen from the Department of Microbiology and Immunology and the Infectious Diseases Translational Research Program at the NUS Yong Loo Lin School of Medicine (NUS Medicine), the insights could help scientists to better understand how pathogenic bacteria evolve and become increasingly virulent and resistant to antibiotics.
The ability to share genetic material is the major driver of microbial evolution because it can transform a benign bacterium into a deadly pathogen in an instant. Phages, the viruses of bacteria, can act as conduits that allow genes to transfer from one bacterium to another by a process known as genetic transduction.
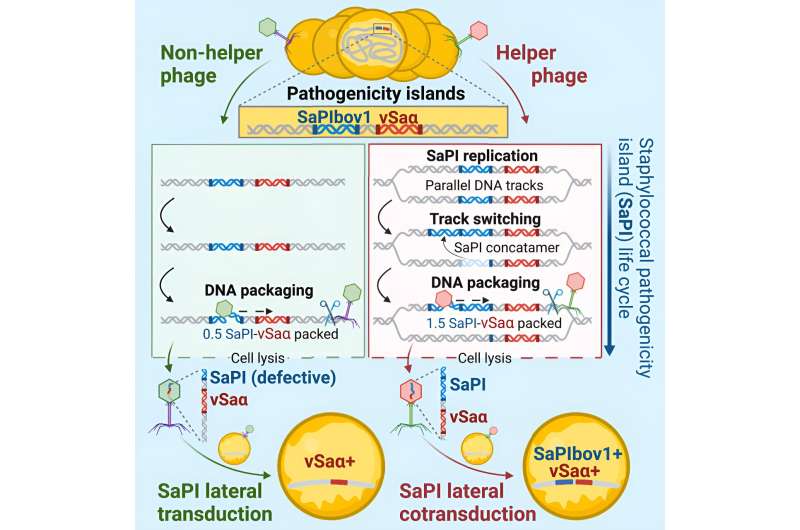
Currently, there are three known mechanisms of transduction: generalized, specialized, and lateral. Lateral transduction was also discovered by the same groups of researchers in 2018, and it is at least one thousand times more efficient than the next most powerful mechanism, generalized transduction.
The new process is termed lateral cotransduction, and the architects behind this new frequency and speed in bacterial evolution are the Staphylococcus aureus pathogenicity islands (SaPIs), which are selfish DNA elements that exploit and parasitise phages and are commonly found integrated into the chromosomes of S. aureus isolates.
S. aureus is a type of bacteria that can cause Staph infections in humans and animals. While it primarily manifests as skin infections, it can become life-threatening if it spreads to the bloodstream and infects organs, bones, or joints.
Professor José R. Penadés from the Department of Infectious Diseases, and Director for the Center for Bacterial Resistance Biology at Imperial College London, said, "This breakthrough sheds light on a novel pathway through which bacteria evolve. Given the alarming surge of antibiotic-resistant superbugs, comprehending the mechanisms driving bacterial evolution becomes increasingly critical."
This newly-discovered process, lateral cotransduction, rivals lateral transduction in terms of efficiency but surpasses the latter in versatility and complexity. While lateral transduction is only known to occur when dormant phages within bacterial genomes become reactivated and initiate reproduction in the lytic cycle, lateral cotransduction can occur during the reactivation process and the infection of new bacterial cells.
Additionally, unlike phages that sacrifice their genes to transmit bacterial host DNA, SaPIs can transfer themselves completely intact with bacterial DNA through lateral cotransduction. This remarkable capability enables them to perpetually repeat the process, making them significantly more potent and efficient in transmitting bacterial genes.
Asst Prof Chen said, "Through the study, we have demonstrated that bacteria can evolve much faster than we understood. While genetic transduction has always been the exclusive domain of phages, in an unexpected twist of irony, our research has shown that parasites of the most prolific parasites on the planet (the phages) are probably the most powerful and efficient transducing agents currently known."
Prof Chng Wee Joo, Vice-Dean at NUS Medicine, said, "This groundbreaking discovery will impact the way we understand how bacteria evolves through gene transfer, and their potential implications on bacterial infections and diseases. This research is also paramount in informing safe treatment decisions in clinical settings, and it is an absolute honor to have our work published in this prestigious journal."
The rise of superbugs has called for new ways to treat antibiotic-resistant strains. One such method that has gained traction in recent years is phage therapy, which involves the use of phages to eliminate harmful bacteria in infections and diseases. However, instead of just fighting bacteria, some therapeutic phages could turn out to be the unwitting accomplices of SaPIs or other related elements capable of lateral cotransduction.
According to Prof Penadés, "This process likely occurs in various other bacterial species as well. This groundbreaking finding marks a paradigm shift in our understanding of bacterial evolution and will immensely influence the ways we combat antibiotic resistance."
"They [phages] could be used to destroy bacteria in the short term but end up spreading harmful genes to other cells in the long term, which could prove to be disastrous. With this new way of understanding the evolutionary mechanisms of disease-causing organisms, it is important for therapeutic phages to be carefully vetted before they are used for therapy," said Chen.
More information: Melissa Su Juan Chee et al, Dual pathogenicity island transfer by piggybacking lateral transduction, Cell (2023). DOI: 10.1016/j.cell.2023.07.001
Journal information: Cell
Provided by National University of Singapore