This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Noncoding DNA explains a majority of the heritability of dairy cattle traits, like milk production and fertility: Study

Regulatory genes—genes that control how other genes are used—are responsible for 69% of the heritability of dairy cattle traits such as milk production and fertility, according to a study published August 23 in the journal Cell Genomics.
This contribution is 44% more than expected and much higher than previous studies of regulatory genes in humans. The findings, reported by a team of animal and human geneticists, could improve the efficiency of agricultural breeding programs. The study also helps solve the longstanding mystery of why mammalian genomes contain so much noncoding DNA.
"We suspect that our large sample size is one of the major reasons why we see that mutations affecting gene expression and RNA splicing are playing a major role in shaping traits," says computational scientist and first author Ruidong Xiang of the University of Melbourne, Agriculture Victoria, and the Baker Heart and Diabetes Institute. "Biomedical science is heavily invested in human genomics, so I think it's a good thing to show that similar and valuable work is happening in other species as well."
Most of our genome is made up of "noncoding" DNA that does not code for protein, but the exact function of these noncoding regions remains something of a mystery. Because many genes in these noncoding regions are associated with specific traits like body size or hair color, scientists have long suspected that these genes contribute to an individual's physical features or "phenotype" by regulating how other genes are used.
However, up until now, there has been limited evidence to support a large role for regulatory genes in determining traits. For example, a recent human study estimated that only 11% of trait heritability is attributable to regulatory genes that alter gene expression.
"This creates a paradox called 'missing regulation," says Xiang. "We decided to look at this problem to see how much variation in cattle traits can be explained by mutations changing gene expression or RNA splicing."
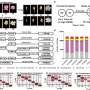
To do this, the team first used the Cattle Genotype-Tissue Expression (CattleGTEx) atlas to build a model of regulatory genes including both gene expression and RNA splicing genes. Then, they used their model to quantify how mutations in these regulatory genes impacted the heritability of traits in a separate dataset comprised of more than 120,000 dairy cattle genomes. Altogether, they examined 37 traits related to milk production, mastitis, fertility, temperament, and body size.
The researchers found that regulatory genes were responsible for 69% of trait heritability, as an average across 37 analyzed traits. "We were surprised, but we performed several validation tests, and the results were consistent, so we began to be convinced that this is the answer, at least in cattle," says Xiang.
Unlike most prior studies that only examined variants of gene expression, this study simultaneously examined both gene expression and RNA splicing genes. They also examined both 'cis' and 'trans' variants—mutations that are located close or far away, respectively, from the coding region that they impact. Most other studies have focused only on the effects of nearby 'cis' mutations, but the researchers say that including 'trans' mutations in the analysis provided another boost to their analysis' power that probably contributed to this study's surprising result.
"Trans effects still haven't been well studied, but we showed that these distant regulatory genes also play an important part in shaping mammalian traits—it appears to be less important than cis effects, but it is important," says Xiang.
These findings could be a boon to farmers looking to selectively breed certain cattle traits. "By mapping genetic variants related to gene expression and RNA splicing, we can map the majority of the heritability of traits," says Xiang. "Therefore, basic knowledge of the mechanisms behind mutations can be used to enhance applications such as breeding in agriculture."
Beyond agriculture, this study helps by providing a model system for future studies in humans and other animals. "The next step is to use this information to try and generate better predictive models for different traits," says Xiang. "Our methods could also be used as a model for other species."
More information: Ruidong Xiang, Gene expression and RNA splicing explain large proportions of the heritability for complex traits in cattle, Cell Genomics (2023). DOI: 10.1016/j.xgen.2023.100385. www.cell.com/cell-genomics/ful … 2666-979X(23)00182-9
Journal information: Cell Genomics
Provided by Cell Press