This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Study discovers novel region for BRD4 transcription and potential therapeutic target
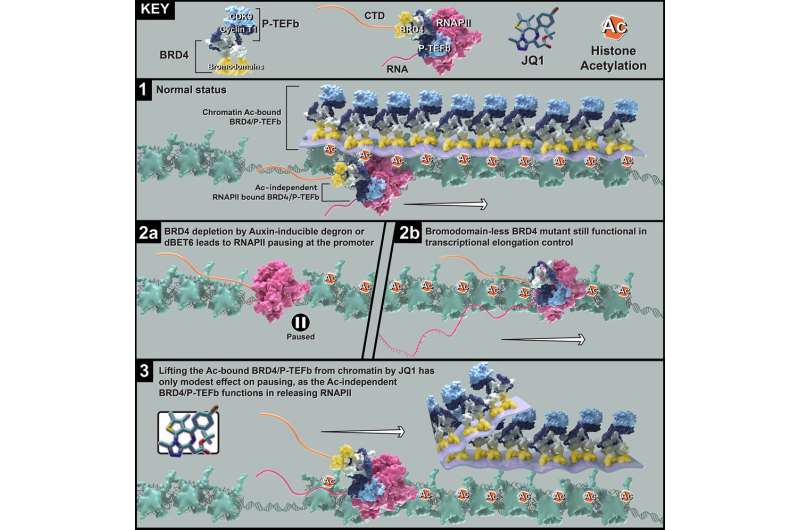
A team of investigators in the laboratory of Ali Shilatifard, Ph.D., the Robert Francis Furchgott Professor and chair of Biochemistry and Molecular Genetics, have discovered a novel protein region responsible for regulating DNA transcription elongation.
The findings, published in Molecular Cell, point to a new therapeutic target that could spur the development of more effective therapies for cancer and developmental disorders.
During transcription elongation—the process of synthesizing RNA from DNA—the RNA polymerase II complex, a multiprotein complex, travels down a strand of DNA and copies genetic information from the DNA to a strand of RNA.
Previous work has identified BRD4, a member of the BET (Bromodomain and Extra-Terminal motif) protein family, as a major factor in transcription regulation and RNA polymerase elongation. Dysregulation of BET proteins, especially BRD4, have also been identified as drivers of multiple types of cancer.
In response, many cancer drugs have been designed as BET protein inhibitors to target and remove BRD4 protein, or bromodomains, from chromatin to regulate transcription elongation. However, these drugs have failed to improve patient outcomes with no clear explanation.
"The effort to develop the BET protein inhibitor has been mainly focused on bromodomains, bromodomain 1 and bromodomain 2, for the past decade. Small molecules that target both bromodomains, such as JQ1, have been in monotherapy cancer trials for a while but with only modest efficacy. Although inhibitors that target a specific bromodomain have been achieved, specific inhibition of BRD4 is currently unavailable," said Bin Zheng, Ph.D., a postdoctoral fellow in the Shilatifard laboratory and lead author of the study.
In the current study, using a BRD4 degron, a technique essential for regulating protein degradation, the investigators demonstrated that modulating BRD4 bromodomain 1 and bromodomain 2 resulted in the same phenotype, suggesting that a different domain of BRD4 regulates transcription elongation instead.
"We've demonstrated that BRD4 is not the be-all and end-all as everyone thought it was," said Shilatifard, who is also the director of the Simpson Querrey Institute for Epigenetics and a professor of Pediatrics. "What we showed is that when the BET domain inhibitor binds here and is taken off chromatin, it surprisingly had no effect."
Instead, they discovered that an entirely new protein domain of BRD4, called the C-terminal domain, is essential for regulating this process. Essentially, the C-terminal domain helps release a previously paused RNA Polymerase II, helping it to resume its journey down the strand of DNA and complete transcription elongation.
"Our study provides the rationale and importance to shift the focus from the bromodomains to the C-terminal region of BRD4 to achieve specific and highly potent BRD4 inhibition to control transcription, and hopefully improve the efficacy of the BRD4 inhibitor for cancer therapy," Zheng said.
According to Shilatifard, the team is now focused on identifying molecular inhibitors that regulate the C-terminal domain.
"Our future work will be focusing on the identification of small molecular inhibitors targeting the novel protein domain we have identified within BRD4, and hopefully a new drug that will provide additional strategy for cancer therapy," said Lu Wang, Ph.D., assistant professor of Biochemistry and Molecular Genetics and co-corresponding author of the study.
More information: Bin Zheng et al, Distinct layers of BRD4-PTEFb reveal bromodomain-independent function in transcriptional regulation, Molecular Cell (2023). DOI: 10.1016/j.molcel.2023.06.032
Journal information: Molecular Cell
Provided by Northwestern University