This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Breaking barriers in drug delivery with better lipid nanoparticles
Many diseases can be successfully treated in the simple environment of a cell culture dish, but to successfully treat real people, the drug agent has to take a journey through the infinitely more complex environment within our bodies and arrive, intact, inside the affected cells. This process, called drug delivery, is one of the most significant barriers in medicine.
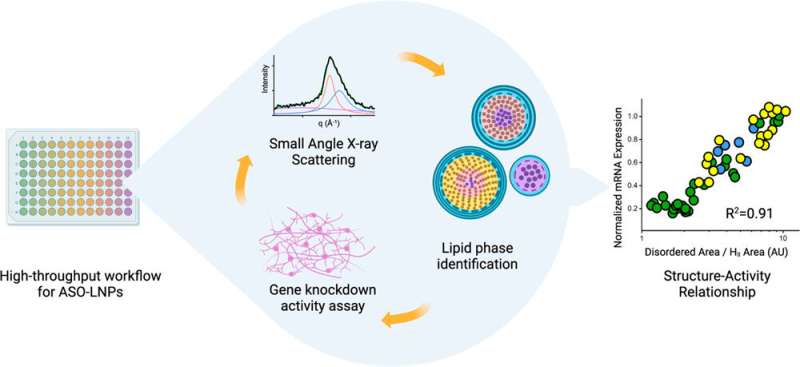
A collaboration between Lawrence Berkeley National Laboratory (Berkeley Lab) and Genentech, a member of the Roche Group, is working to break through some of the drug delivery bottlenecks by designing the most effective lipid nanoparticles (LNPs)—tiny spherical pouches made of fatty molecules that encapsulate therapeutic agents until they dock with cell membranes and release their contents. The first drug to use LNPs was approved in 2018, but the delivery method rose to global prominence with the Pfizer and Moderna mRNA COVID vaccines.
"It's quite a smart system, because if you just deliver the RNA itself to the human body, the RNA is degraded by nucleases and cannot easily cross the cell membrane due to its size and charge, but the LNPs deliver it safely into the cell," explained co-lead author Chun-Wan Yen, a senior Principal Scientist in Genentech's Small Molecule Pharmaceutical Sciences group.
LNPs are now being widely explored as a delivery system for vaccines for other infectious diseases or therapeutic vaccines for cancer. The viability of these new applications will be dependent on how well the lipid envelopes fuse with target cells, how stable the drug-LNP formulations are in storage (so that they have a long shelf-life), and how stable they are in the body (so they can confer prolonged drug activity).
All these properties are controlled by the mixture of molecules used to create the LNP, and the resulting 3D structure of the particle. The team under Yen and fellow co-leads Greg Hura and Michal Hammel, both Berkeley Lab biophysicists, has been studying how to tune the structure of LNPs for desired properties for several years.
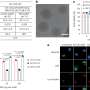
Their latest paper, published recently in ACS Nano, documents how a high-throughput workflow allows them to produce and characterize LNPs at record speed. The study also includes the first-ever demonstration of how LNP structure correlates with the activity of its contents, which for this investigation was an anti-sense oligonucleotide (ASO). ASOs are small snippets of RNA or DNA base pairs that block gene expression by binding to strands of mRNA and preventing them from being translated into proteins. ASOs are a great way to treat diseases caused by faulty proteins or the over-abundance of a protein. But, like mRNA, they are susceptible to roving nucleases—enzymes that degrade RNA and DNA—and cells do not readily uptake them.
The scientists discovered that ASO-carrying LNPs with neatly ordered, closely-packed internal structures led to better silencing of a faulty gene in human neurons that is associated with a degenerative disease, compared with LNPs that had a more disordered structure. The findings were from cellular activities, not from animal studies, so there is still more work ahead, but the team are excited to build upon these insights using the complementary tools of each institution.

"We generate the LNPs in high-throughput and Greg and Michal's team can offer the high-throughput analysis," said Yen. "If you check about the publication nowadays, they typically just do one or two formulations, but for us it's different. We can generate large datasets, and I think that's the reason why we can have this very unique and cool finding."
"This paper really sets out the method that we're going to apply to the thousands of other formulations that we intend to characterize," added Hura, who is part of Berkeley Lab's Biosciences Area. "We hope that this will be a general method for people to optimize their lipid nanoparticles. Whether that be vaccines, and again, the vaccines have received the greatest attention so far, or beyond. This type of therapy has a very broad application beyond that."
How to build a lipid nanoparticle
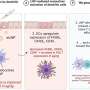
The structures of LNPs are affected by how you mix them, what you mix together, and in what order. LNPs have four ingredients—ionizable lipids, helper phospholipids, cholesterol, and polyethylene glycol-lipids (PEG-lipids)—and each ingredient has different forms. Plus, they can be combined in different ratios, leading to an exponential number of possible formulas. Further complicating matters, the LNPs change with time. A formulation that begins as a neat, close-packed sphere will morph into a more disordered structure eventually.
Scientists at Genentech developed a robot-driven workflow that can generate hundreds of LNP formulations in just a few hours. Samples of each formulation are then brought to Berkeley Lab to perform small-angle X-ray scattering (SAXS) at the Advanced Light Source, a circular particle accelerator that creates X-ray beams of different energies.
The biological SAXS beamline can quickly process many samples, and unlike other forms of X-ray diffraction on biological materials, the samples do not need to be frozen or crystalized—which could change the structure of the LNPs and prevent the scientists from discovering what the LNPs would look like at physiological temperatures in the human body. SAXS also allows them to take snapshots of LNPs at a specified timepoints to determine their structural longevity.
Additionally, the Genentech team uses an accelerated process to study how LNPs affect gene expression in their target cells. By combining all these expedited techniques, the entire collaboration is able to screen potential LNPs at an unprecedented rate.
Yen plans to continue using the SAXS beamline to study small details, like how a 1% change in ingredient concentration or using a new machine during production can affect LNP's cellular activity, as well as big questions, such as whether LNPs behave differently if they are carrying other cargo types and how they interact with different target cells.
"We know that mRNA LNPs work, but there is still a huge knowledge gap," Yen said. "That's why I feel like our paper is a pioneer in this field and hopefully we can also generate more data and understanding for the future applications."
More information: Michal Hammel et al, Correlating the Structure and Gene Silencing Activity of Oligonucleotide-Loaded Lipid Nanoparticles Using Small-Angle X-ray Scattering, ACS Nano (2023). DOI: 10.1021/acsnano.3c01186
Journal information: ACS Nano
Provided by Lawrence Berkeley National Laboratory