This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Team develops organic redox polymer for aluminum-ion batteries with improved storage capacity
Aluminum-ion batteries are seen as a promising alternative to conventional batteries that use scarce and difficult-to-recycle raw materials such as lithium. This is because aluminum is one of the most common elements in the Earth's crust, is easier to recycle and is also safer and less expensive than lithium. However, the development of such aluminum-ion batteries is still in its infancy, as suitable electrode materials that provide sufficient storage capacity are still lacking.
A research team headed by Gauthier Studer and led by Prof. Dr. Birgit Esser of the University of Ulm and Prof. Dr. Ingo Krossing as well as Prof. Dr. Anna Fischer of the University of Freiburg has now developed a positive electrode material consisting of an organic redox polymer based on phenothiazine. In the experiment, aluminum batteries with this electrode material stored a previously unattained capacity of 167 milliampere hours per gram (mAh/g). The organic redox polymer thus surpasses the capacity of graphite, which has mostly been used as an electrode material in batteries to date. The results are published in the journal Energy & Environmental Science.
Electrode material inserts complex aluminum anions
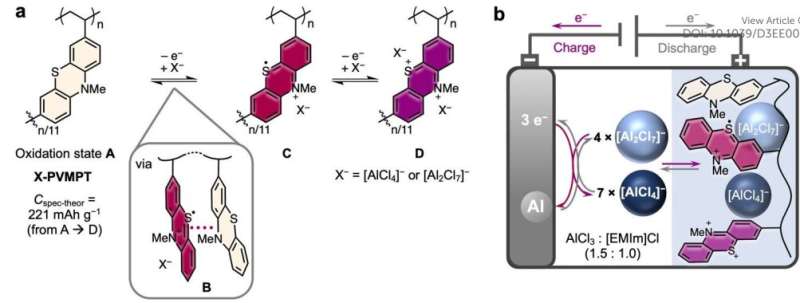
The electrode material is oxidized during charging of the battery, thereby taking up complex aluminate anions. In this way, the organic redox polymer poly(3-vinyl-N-methylphenothiazine) manages to insert two [AlCl4]− anions reversibly during charging. The researchers used the ionic liquid ethylmethylimidazolium chloride as electrolyte with added aluminum chloride.
"The study of aluminum batteries is an exciting field of research with great potential for future energy storage systems," says Gauthier Studer. "Our focus lies on developing new organic redox-active materials that exhibit high performance and reversible properties. By studying the redox properties of poly(3-vinyl-N-methylphenothiazine) in chloroaluminate-based ionic liquid, we have made a significant breakthrough by demonstrating for the first time a reversible two-electron redox process for a phenothiazine-based electrode material."
Poly(3-vinyl-N-methylphenothiazine) deposits the [AlCl4]− anions at potentials of 0.81 and 1.65 volts and provides specific capacities of up to 167 mAh/g. In contrast, the discharge capacity of graphite as electrode material in aluminum batteries is 120 mAh/g. After 5,000 charge cycles, the battery presented by the research team still has 88% of its capacity at 10 C, i.e., at a charge and discharge rate of six minutes. At a lower C rate, i.e., a longer charge and discharge time, the battery returns unchanged to its original capacities.
"With its high discharge voltage and specific capacity, as well as its excellent capacity retention at fast C rates, the electrode material represents a major advance in the development of rechargeable aluminum batteries and thus of advanced and affordable energy storage solutions," says Birgit Esser.
More information: Gauthier Studer et al, On a high-capacity aluminium battery with a two-electron phenothiazine redox polymer as positive electrode, Energy & Environmental Science (2023). DOI: 10.1039/D3EE00235G
Journal information: Energy & Environmental Science
Provided by University of Freiburg