This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
proofread
Exploring how to develop better rechargeable aluminum batteries
A team from China published new work on rechargeable aluminum batteries in Energy Material Advances.
"Rechargeable aluminum batteries (RABs) have great potential as powerful candidates for large-scale energy storage devices," said the corresponding author Chuan Wu, professor at School of Materials Science and Engineering in Beijing Institute of Technology. "The high theoretical capacity, abundant reserves and high security will help RABs achieve application and commercialization."
Professor Wu explained that due to the multi-electron reaction of Al (three-electron-transfer in theory), the direct adoption of Al anode could lead to highly competitive gravimetric and volumetric capacities (2980 mAh g−1 and 8046 mAh cm−3 based on Al) as well as suitable redox potential (−1.66 V versus standard hydrogen for Al3+/Al). However, stemming from the basic properties of a small radius (54 pm) and high charge (+3), Al3+ ions exhibit extremely slow migration activity and strong electrostatic force to damage the crystal structure if migrated.
These intrinsic characteristics lead to low open-circuit voltage, capacity fading and unsatisfactory cycling performance, hindering the applications of RABs. Furthermore, side reaction of electrolyte and Al anode corrosion are also urgent issues for researchers to solve in order to get excellent performance of RABs. Accordingly, it is crucial to understand the reaction mechanism and accordingly design the cathode materials.
2-dimensional (2D) materials attracted researchers' sights owing to tunable electronic states, ion-hosting ability and certain special properties, exemplified by graphene, MoS2, phospholene, silicene, etc. To explore the suitable cathode materials, Wu and his team paid attention to the borophene which was first synthesized in 2015.
According to the reports in battery application, the reaction voltages (0.15 to 0.96 V against Li, Na, Mg, K, and Ca metals) are almost suitable as anode materials, and the diffusion energy barriers of charge carriers are low (325 meV for Li, 3 meV for Na, 28 meV for Mg, 440 meV for Ca, and 4 meV for K). Therefore, this implies the superior electrochemical performances of borophene that can be a potential electrode material for aluminum battery, especially plagued by diffusion kinetics.
To understand the specific Al-ion hosting properties of borophene, Wu and his team have investigated the crystal structure, electronic structure, equilibrium voltage and ions diffusion kinetics as electrode material for RABs.
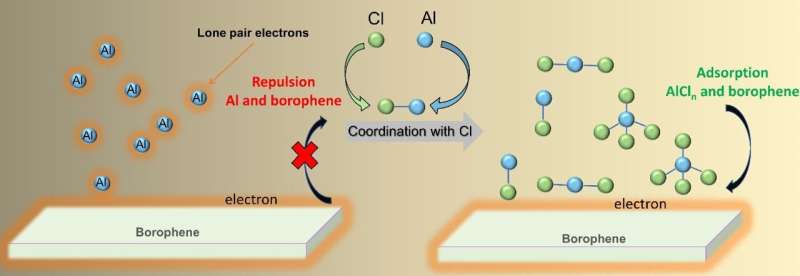
"Different from a traditional recognition, the electrolyte environment in RABs actually has profound influence on the theory redox potential because the Al-anion complex can act as carrier and, hence, the change of reaction occurs. Typically, in ionic liquid electrolyte or ionic liquid analogs (best option for Al anode owing to the compatibility and high deposition/dissolution efficiency), except for Al3+, the anionic species AlCl4− and Al2Cl7− and cationic species AlCl2+ and AlCl2+ can exist in the form of Al complex," Wu said.
"Hence, it would be necessary to revisit the electrochemical performance of borophene considering the effect of AlCln complex ions and to understand the favored reaction mechanism."
It is found that, "After comparing the adsorption of Cl−, Li+, and Al3+ ions, the electron transfer reaction is hindered by the strong Coulomb interaction in the process of Al3+ adsorption. The origin lies at the stable 3s orbital and the large gap between 3s and Fermi level to forbid the further electron transfer of Al," Wu said. "The lone pair electrons of 3s orbital are repulsive to the electron-gaining borophene."
"When we take AlCl4−, AlCl2+, and AlCl2+ ions as carrier in RABs into consideration systematically, we find that because of the coordination of Cl to Al, the multielectron transfer is unlocked accordingly: The gap between 3s and Fermi level decreases after orbital hybridization, and the electron transition in 3s orbital is easy to occur for AlCln," Wu said.
"Coordination with Cl (as AlCln) decreases the gap between Al 3s orbital and Fermi level to assist electron transfer."
"The high capacities of 490 mAh/g ([AlCl2]0.17B) and 841 mAh/g ([AlCl]0.33B) are predicted in the DFT study with the average voltages of 2.037 V ([AlCl2]0.17B) and 1.018 V ([AlCl]0.33B) against Al. And the AlCln diffusions on the borophene are 0.46 eV for AlCl4, 0.20 eV for AlCl2, and 0.08 eV for AlCl, respectively," Wu said.
"The fast kinetics of diffusion, high storage capacity, and voltage indicates a superior electrochemical performance of borophene in RABs."
This work maps out a possibly universal role of Cl coordination in fulfilling multielectron reaction and shed light upon the complicated multicarrier (AlCln) redox condition of RABs. Wu said, such conclusions can predict the performance of borophene as electrode materials and provide an important viewpoint for the multicarrier in the case of RABs.
"Although great progress has been achieved, the development of Al cathodes for RABs is still facing massive challenges, such as slow ions dynamics of Al3+ and complex reaction mechanism of Al and AlCln," Wu said.
"In practical application, RABs need more attention to decrease the side reaction, delay Al anode corrosion and explore new cathode materials. If the new materials, especially aluminum-containing cathode materials, could achieve a high energy density and long cycle life, RABs will become an integral part of electrochemical energy storage. In a word, achieving the commercialization of RABs requires more attention to solve more practical issues."
More information: Lumin Zheng et al, Multielectron reaction of AlCl n in borophene for rechargeable aluminum batteries, Energy Material Advances (2022). DOI: 10.34133/energymatadv.0005
Provided by Beijing Institute of Technology Press Co., Ltd