This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Uncovering the secrets of how chromosomes assemble
Experiments using extracts from African clawed frog eggs have revealed how a key protein complex is regulated to assemble chromosomes during cell division. This finding could help to shed light on the development of certain cancers and birth defects that result when this process goes awry. The findings are published in the journal eLife.
In preparation for cell division, DNA is replicated, and the replicated DNA is packaged into an X-shaped chromosome. This process is essential to ensure that each daughter cell receives a complete copy of the genetic material from the parent cell during a process called mitosis.
A protein complex known as condensin I plays a key role in folding DNA to assemble chromosomes during mitosis. It remained unclear, however, exactly how the activity of condensin I was regulated.
"The activity of condensin I must be tightly regulated during the cell cycle," says Tatsuya Hirano of the RIKEN Chromosome Dynamics Laboratory. "If condensin I was prematurely activated before the entry into mitosis, for instance, it could damage DNA or cause chromosomes to become unstable, potentially producing cancer cells."
In previous experiments, the team found that condensin I is activated during mitosis through a process called phosphorylation in which many phosphoryl groups (PO3) are added to its subunits. However, condensin I is a large protein complex with five subunits, and it was unclear which site or sites within are phosphorylated during mitosis, and how this affects the overall function of the protein complex.
To explore this, Hirano's team has used an experimental system they developed many years ago—a powerful functional assay based on African clawed frog egg extracts. These extracts contain all components required for assembling chromosomes, including condensin I.
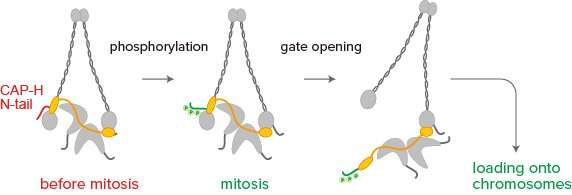
In the new study, the team focused on the N-terminal tail (or N-tail) of the subunit CAP-H, and tested what would happen when existing condensin I in the extracts was replaced by its mutant forms.
The researchers found that deletion of the N-tail accelerated condensin I loading and chromosome assembly, and gathered evidence that phosphorylation of the CAP-H N-tail promotes condensin I loading onto chromosomes. They propose that the N-tail may act as a gatekeeper of condensin I for its action during chromosome assembly.
The team was also surprised to find that, when the N-tail was deleted or compromised, condensin I could trigger the assembly of chromosome-like structures even without proper phosphorylation. This suggests that, under certain circumstances, condensin I may be able to bypass the need for phosphorylation.
"This study represents one of the successful outcomes of a decade of persistent efforts in our lab," says Hirano. "We hope to fully understand the mechanism of condensin I activation in further research."
More information: Shoji Tane et al, Cell cycle-specific loading of condensin I is regulated by the N-terminal tail of its kleisin subunit, eLife (2022). DOI: 10.7554/eLife.84694
Journal information: eLife
Provided by RIKEN