This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Cell death, a life-giving event, can also trigger severe disease

When the body machinery that kills off hundreds of millions of cells a day fails, inflammation and sickness are often not far behind.
Cell death, which might sound unwelcome, is actually essential for keeping every person alive.
The process, tightly regulated by the body, destroys old or damaged cells to make way for new ones. At the same time, cells invaded by microbes are eliminated to fight infections.
"Most people don't realize this, but every second there are about 4 million cells dying in each of our bodies to be replaced with fresh ones," said Dr. Mohamed Lamkanfi, an immunologist in the Department of Internal Medicine and Pediatrics at Ghent University in Belgium. "Programmed cell death is a fundamental part of life."
Flawed processes
But the system must work flawlessly for optimal health and sometimes errors occur. These can lead to tissue damage, inflammation and sickness.
For instance, cells might survive beyond their optimal life span and cause an autoimmune disease, stubborn viral infections or even cancer. At the other end of the spectrum, excessive cell death could result in tissue degeneration and cause severe disorders such as Alzheimer's and Parkinson's.
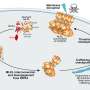
Lamkanfi is particularly interested in pyroptosis, a form of cell death that involves the destruction of important white blood cells known as macrophages, which usually kill invading microorganisms and stimulate other parts of the immune system.
Pyroptosis causes inflammation and, while an inflammatory state is crucial when a person is fighting an infection, it can also be highly detrimental when it happens at the wrong time or in excess. Uncontrolled pyroptosis is linked to chronic inflammatory conditions such as rheumatoid arthritis and familial Mediterranean fever (FMF).
Lamkanfi was principal investigator on a project called PyroPop and has a particular interest in FMF, an inherited disorder that usually occurs in people of Mediterranean and Middle Eastern origin, because he has family ties to Morocco.
Personal interest
"You might say that my interest in familial Mediterranean fever is personal," said Lamkanfi. "My family originates from Morocco, where the disease is endemic and a huge health burden for many patients."
In countries where FMF is endemic, between one in 400 and one in 1,000 people is affected by the disease and there are clinics in parts of the Mediterranean dedicated entirely to treating it. Severe bouts of FMF-induced fever can lead to organ damage, joint pain and infertility, among other complications.
Through his work on PyroPop, which was completed in 2021, Lamkanfi has been better able to understand the complex sequence of cellular events that give rise to pyroptosis.
A follow-up project called PyroScreen is now under way. The aim of this new research is to find treatments to stave off inflammation.
Lamkanfi is not alone in his quest for treatments to counter the negative effects of cell death. Indeed, finding ways to intervene when programmed cell death goes wrong has been a research priority in many laboratories around the world over the past 20 years and drug trials are now under way.
Researchers are hopeful that remedies will be found within the next 10 years "so we can better treat some very serious diseases," said Dr. Manolis Pasparakis, a professor of genetics at the University of Cologne in Germany.
Natural demise
Since the discovery almost 200 years ago that cell death is a natural part of life, scientists have zeroed in on many key players of the machinery behind the process. They have identified both genes that regulate cell death and the molecules that precipitate the cascade of events resulting in a cell's destruction.
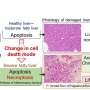
Healthy, programmed cell death is known as apoptosis and enzymes called caspases play an important part in making it happen. Molecules from this family ensure a careful degradation of a dying cell.
This orchestrated collapse includes deformation of the cell membrane, cell shrinkage and fragmentation of the DNA and all the cell's contents. The cell's corpse—shriveled but intact—is then quickly swallowed up by neighboring cells.
The net result is a discrete elimination of a cell that has passed its sell-by date, triggering no immune response from the body.
Exploding cells
The same cannot be said for necroptosis, another tightly regulated form of cell death. There is nothing quiet or discrete about it.
"Cells undergoing necroptosis rupture and dump their contents into the space between cells and this becomes dangerous," said Pasparakis. "The exploded contents act on receptors on neighboring cells and induce inflammation and tissue damage."
He is principal investigator of the Necroptosis project, which is exploring the role of cell death in immunity and inflammation. The initiative, which began in 2018, runs through September this year.
In 2009, Pasparakis and his team discovered the pro-inflammatory properties of necroptosis by accident while studying genetically modified mice.
"We expected to have mice that would be resistant to cell death, but we found the opposite," he said. "The mice developed a very severe inflammatory response, causing sickness and death."
Today, the pros and cons of necroptosis are better understood.
"We know it's important in the body's defense against viruses: a cell must die fast when it's infected by a virus, before the virus has a chance to replicate, and necroptosis is a great way to facilitate a quick death," Pasparakis said. "On the other hand, necroptosis is also a highly inflammatory type of cell death, so when it happens in excess it can trigger severe disease."
While evidence that this is the case comes from mouse studies, whether necroptosis causes human disease has yet to be confirmed. Proof may come from clinical trials happening now.
"The whole science community is waiting anxiously to see the results," said Pasparakis.
Positive results promise to revolutionize the treatment of many debilitating diseases.
"Once we've found a way to block the faulty cell-death machinery, we should see an impressive clearing of pathology [sickness] from the body in certain inflammatory diseases," said Lamkanfi at Ghent University. "We have already seen this in mice."
By extension, that could mean "new therapies and diagnostics for millions of people suffering from chronic inflammatory diseases, from gout and heart disease to neurodegenerative conditions," he said. "The impact could be remarkable."
Provided by Horizon: The EU Research & Innovation Magazine