This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Researchers realize selective recovery of high-value rare earth elements from waste NdFeB

Recycling high-value rare earth elements from NdFeB waste can save resources, reduce industrial waste, protect the environment and bring considerable economic benefits. However, most traditional recovery methods take a long time to process, have high acid consumption, and low selectivity.
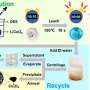
In a study published in Separation and Purification Technology, the research group led by Prof. Yang Fan from Fujian Institute of Research on the Structure of Matter of the Chinese Academy of Sciences proposed a new integrated method for recycling permanent magnet waste by betaine hydrochloride ([Hbet]Cl) solution extraction.
The researchers first carried out leaching experiments on fully roasted magnetic clay samples, and selected [Hbet]Cl as the leaching agent.
They found that when leaching temperature was 200° Celsius, leaching time was eight hours, lixiviant [Hbet]Cl concentration was 0.2 mol/L, and solid–liquid ratio was 1:150 g/ml. They were able to get optimum leaching results of 99.81% Pr, 97.05% Nd, 95.51% Gd, 56.24% Ce and 0.20% Fe, and almost all iron oxide remained in the residue.
Compared with methods for common lixiviants, the results of the proposed method showed a better leaching rate and selectivity. And the leaching sequence was in line with established chemical properties.
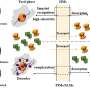
In addition, the researchers revealed that the new lixiviant ([Hbet]Cl) will not reduce the extraction rate of rare earth materials by N, N-di-2-ethylhexyl diglycolamic acid (D2EHDGAA) extractant, and the extraction rate of D2EHDGAA for Nd in [Hbet]Cl leaching solution is significantly improved.
More information: Chang Du et al, Recovery of high-value rare earth elements from waste NdFeB by the water-soluble ammonium salt [Hbet]cl, Separation and Purification Technology (2022). DOI: 10.1016/j.seppur.2022.122946
Provided by Chinese Academy of Sciences