This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researchers realize paired electrosynthesis of dimethyl carbonate with single-atom catalyst
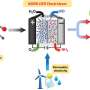
Typically in a traditional electrochemical cell with a membrane separator, the products of cathodic CO2 reduction reaction (CO2RR) are usually in reduced forms. The anodic O2 evolution does not play a role in further expanding the product scope.
Dimethyl carbonate (DMC, CH3OCOOCH3) is an environmental-benign chemical feedstock. The convergent paired electrosynthesis of DMC represents an important revolution for the DMC production.
In a study published in Energy & Environmental Science, a research group led by Prof. Zhu Qilong's group from Fujian Institute of Research on the Structure of Matter of the Chinese Academy of Sciences reported an Ni single-atom catalyst (Ni SAC) for CO2-to-CO conversion and the convergent paired electrosynthesis of DMC.
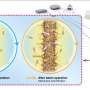
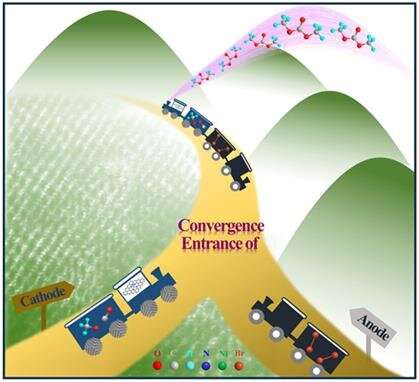
The researchers found that the convergent paired electrosynthesis couples the anodic halide ion oxidation reaction and the cathodic CO2RR to generate the key intermediates for the DMC synthesis. During this conversion, the efficient cathodic CO2-to-CO conversion plays a dominant role, inspiring great effort to control the micro-nanostructure of electrocatalysts.
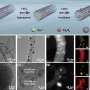
They also revealed that the dual-channel superstructured Ni SAC with a unique site coordination configuration bonded via one axial oxygen atom and four planar nitrogen atoms is controllably constructed and is capable of providing a preeminent performance for CO2-to-CO conversion, achieving the exclusively high Faradaic efficiency (FE) and partial current densities with excellent stability.
By virtue of the atomic to nanoscopic to microscopic manipulation of the penta-coordinated Ni SAC for CO production, the convergent paired electrosynthesis of DMC from CO2 was pioneeringly performed, realizing the high FE of DMC.
The mechanism study unveiled that such axial oxygen coordination configuration is helpful to decrease the energy barriers for the generation of key *COOH intermediate and the dissociation of H2O and CH3OH, accelerating the convergent paired electrosynthesis.
This study suggests that the proof of concept in the innovative convergent paired electrosynthesis could open up a new horizon in the fields of CO2 utilization.
More information: Xiaofang Li et al, Convergent paired electrosynthesis of dimethyl carbonate from carbon dioxide enabled by designing the superstructure of axial oxygen coordinated nickel single-atom catalysts, Energy & Environmental Science (2022). DOI: 10.1039/D2EE03022E
Journal information: Energy & Environmental Science
Provided by Chinese Academy of Sciences