New catalyst proven efficient in electrosynthesis of ammonia
In recent research, researchers led by Prof. Zhang Haimin from the Institute of Solid State Physics of the Hefei Institutes of Physical Science (HFIPS) realized the synthesis of Mo single atoms anchored on activated carbon (Mo-SAs/AC) by the formed Mo-Ox bonds. The result was published in Chemical Communications.
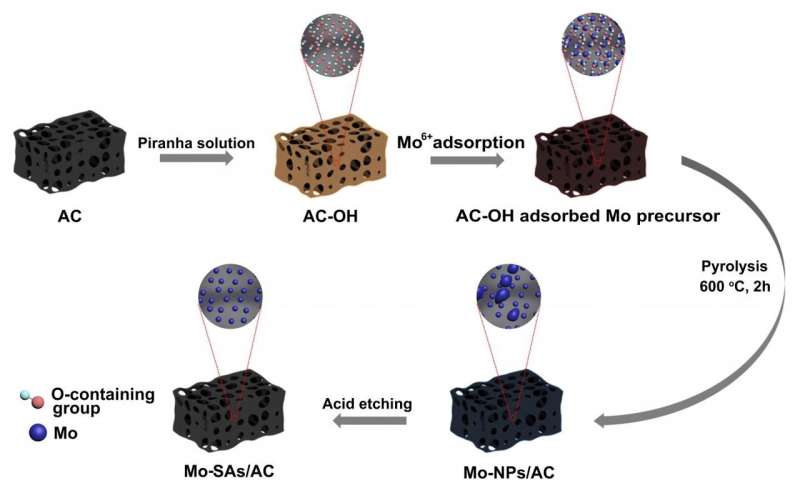
According to the researchers, this new oxygen-coordinated molybdenum single atom catalyst was proved efficient to electrosynthesis of ammonia. The O-coordinated environment in this study, different from N-coordinated environment reported before, provided the sites to anchor Mo single atoms and form Mo-Ox sites, which could be used as the active centers for the adsorption and activation of N2, resulting in high nitrogen reduction reaction (NRR) activity.
"We have been curious about the key to the high NRR catalytic activity," said Geng Jing, first author of the study, "then we found the Mo-Ox site in the catalyst."
In this research, the surface-rich oxygen functional groups of pre-treated activated carbon played an important role in capturing the Mo precursor, forming Mo-O coordination to anchor Mo atoms as the catalytic active sites.
As a result, in a Na2SO4 electrolyte, the Mo-SAs/AC can produce ammonia and attain a faradaic efficiency with high stability and good durability.
This work would be very helpful for designing and developing oxygen-coordinated single atom NRR electrocatalysts for high efficiency electrosynthesis of ammonia.
More information: Jing Geng et al, An oxygen-coordinated molybdenum single atom catalyst for efficient electrosynthesis of ammonia, Chemical Communications (2021). DOI: 10.1039/D1CC01455B
Journal information: Chemical Communications
Provided by Chinese Academy of Sciences