This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Tracing a plant chemistry puzzle to its roots
Graduate student Colin Kim had been making good progress on a scientific puzzle that had fascinated him since he joined Whitehead Institute Member Jing-Ke Weng's lab, trying to understand how coumarin synthase (COSY), an enzyme that plants use to make agriculturally and medicinally important molecules called coumarins, does its job.
A series of successful experiments had illuminated many of COSY's secrets, but these had been followed by unsuccessful experiments. Now Kim found himself in a situation familiar to every research scientist: he was stuck. There was a missing piece in the puzzle of how COSY works, some subtle bit of chemistry that he hadn't been able to identify.
Hoping to come across an idea that might help solve the puzzle, Kim began scouring old research papers related to plant production of coumarins. Scientists constantly refer to past studies to understand what is already known about their research subject, and to hunt for experimental techniques that they might be able to apply to their own work. Weng and Kim hoped that a historical paper might spark such inspiration.
The lightbulb moment struck when Kim was reading a paper from 2008, years before COSY had been discovered. The researchers had performed an odd experiment with cassava roots and made a startling finding; was their insight the clue that Kim needed? Performing a version of the experiment himself would soon reveal the answer.
A puzzle presents itself
Kim's efforts to understand COSY began in 2019, shortly after he joined Weng's lab, when then-postdoctoral researcher in the lab Andrew Mitchell introduced him to the project. Researchers at Ghent University, led by first authors Ruben Vanholme and Lisa Sundin, had just published their discovery of the enzyme.
Enzymes are molecules that enable or speed up chemical reactions, and plants have evolved many specialized enzymes in order to create a wealth of natural molecular products. Because plants cannot move, they instead produce an arsenal of molecules with which they can react to their environments: molecules that protect them against pests and herbivores, maximize their absorption of nutrients, attract pollinators, help them tolerate drought, and more.
Coumarins are a family of molecules produced by many plants, which help them to draw iron out of the soil. Plants also use coumarins as part of their chemical defenses and to help regulate interactions with microbes. Coumarins also have potent medicinal properties. They can be used to prevent blood clots; warfarin, a blood thinner that is one of the most prescribed medications in the U.S., is derived from a coumarin.
Researchers used to think that plants relied on spontaneous chemical reactions, facilitated by light, to achieve important steps in the process of producing coumarins. Then in 2019, Vanholme, Sundin, and colleagues discovered COSY in the model plant Arabidopsis thaliana, and found that plants use the enzyme to speed up coumarin production.
COSY belongs to a family of enzymes called BAHD acyltransferases, but its activity is very different from its close relatives. Other BAHDs take in two molecules and join them together. COSY works on one molecule, and enables changes within that molecule that ultimately enable it to form a ring shape, which is necessary to make the coumarin. COSY does this using a trans-to-cis isomerization, which means it breaks a double bond between two carbons—typically a very stable chemical bond and so takes a lot of energy to break—allowing a chemical group to flip from one side of the double bond to the other side, where the chemical group is used to form the ring.
Kim wanted to know how COSY is able to facilitate this unique chemistry. In order to solve the puzzle, Kim first set out to get a better picture of COSY.
A picture of COSY crystallizes
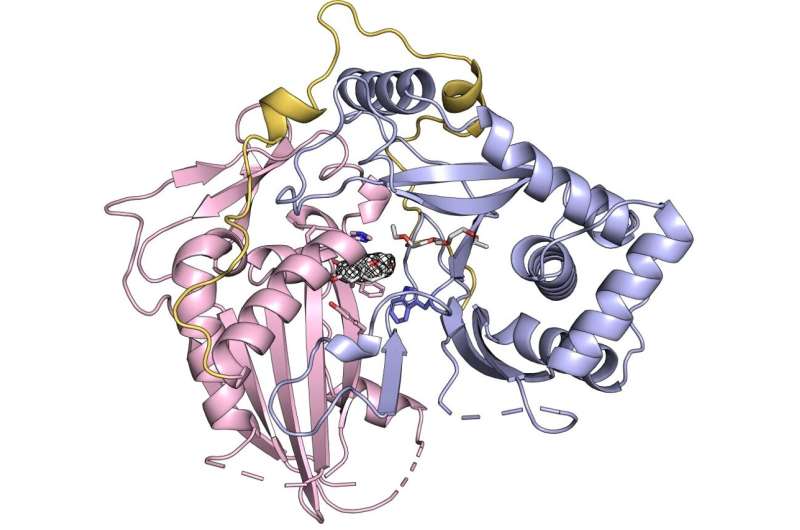
Kim and colleagues used a process called crystallization to capture COSY's 3D structure and molecular makeup. At first glance, COSY appeared very similar to other BAHD acyltransferases. It had the same overall shape: two large globs connected by a stringy section called a linker loop. What makes COSY unique only became apparent when the researchers looked closely at the active site, or location where enzymes interact with the molecules that they affect.
Most BAHDs have two binding pockets in their active site, spots where they take in the two molecules that they interact with. One of COSY's binding pockets has been blocked off, leaving only the other binding pocket available. By losing a binding pocket, COSY adapted to make internal changes to a single molecule, rather than to join two molecules. This discovery helped illuminate how COSY evolved from other BAHDs: a simple restructuring allowed COSY to diverge from its enzyme relatives and assume a completely new function.
With the crystal structure in hand, Kim next wanted to understand how COSY actually interacts with the molecule it turns into a coumarin. He began systematically mutating COSY, eliminating parts of the enzyme, in order to see what parts were necessary. However, the part Kim most expected to be necessary—a histidine, an amino acid that is essential in other BAHDs—turned out to be non-essential to COSY's function. Kim was stumped. It was time to do some homework—reading old papers.
A hint from the past
Kim and colleagues looked to past research on coumarin synthesis in plants. Perhaps, even though past researchers had not known of the existence of COSY, they had nonetheless identified aspects of coumarin production that could provide clues to how COSY works. Kim's search led him to the 2008 paper by Soad A.L. Bayoumi and colleagues at the University of Bath, in which they found that coumarins were accumulating in cassava roots after harvesting.
At the time, researchers thought that the chemical reaction in plants to create coumarins happened spontaneously, facilitated by light. However, this work found that light was not required, and concluded that there might instead be an unknown enzyme playing a role in coumarin synthesis. They reached that conclusion after feeding cassava roots molecules containing deuterium, a variant of hydrogen, in order to find out if the production of coumarins involved acid-base chemistry.
When a plant enzyme carries out acid-base chemistry, this happens invisibly, because the enzyme borrows a hydrogen that immediately gets replaced by an identical hydrogen. However, if researchers get the plant to grow such that its molecules contain deuterium, then they can see if the plant replaces the deuterium with a hydrogen and know that the reaction occurred.
By feeding cassava roots with deuterium-containing precursors to coumarin, Bayoumi et al were able to observe a deuterium-hydrogen swap during coumarin synthesis. This suggested to them that some sort of acid-base chemistry, facilitated by an enzyme, was going on. Kim was sure they were right, and now that the speculative enzyme had been found, he could confirm their hypothesis.
Kim and colleagues set up three variations of hydrogen/deuterium experiments with the crystal-structure of COSY in mind. All three showed that COSY uses its unique active-site geometry to borrow a nearby hydrogen and exchange it onto the molecule that it acts on. An invisible step in COSY's process had been rendered visible. Now that Kim knew COSY used acid-base chemistry, he could figure out how it worked.
Kim and collaborators from the Massachusetts Institute of Technology (MIT)'s department of chemical engineering used molecular dynamics simulations and quantum mechanical modeling to outline, step by step, how COSY catalyzes the necessary changes to the coumarin precursor molecule that it works on.
In the end, this is what they determined, as published in Nature Communications on February 3: COSY takes in one molecule, a precursor to coumarins. It uses acid-base chemistry, borrowing a hydrogen from a nearby water molecule, to convert the precursor molecule's double bond to a single bond. The molecule can then reorientate itself around this rotatable single bond into the right position to form a ring, which leads to the production of a coumarin molecule. Without COSY, the reaction would occur spontaneously through an alternative route, but at a much slower pace.
What COSY's secrets reveal
Solving how COSY works might help in the production of coumarin products in agriculture and medicine. It might also help researchers who are trying to engineer proteins, as they can learn from COSY's structure and mechanisms. But mostly, Weng and Kim feel that solving the puzzle of COSY illustrates the beauty and strength of plant evolution.
"All higher plants contain COSY, and what that tells us is that the spontaneous reaction is not fast enough," says Weng, who is also an associate professor of biology at MIT. "Nature is harsh, and the little edge that plants can get by evolving a new enzyme to accelerate coumarin production is beneficial enough that the plants that can't do that die out."
"On the surface, COSY and its enzyme relatives look the same, but when you actually zoom in, there are subtle changes that completely change the trajectory of the enzyme function, and drive it to carry out this highly specialized and yet widely important task in plants," Kim says.
If COSY had not evolved its single binding pocket, with its unique configuration, then it would not be able to carry out the elaborate acid-base chemistry that gives COSY-containing plants their edge.
"The active site has evolved and restructured to allow for this reaction to take place in a very intricate and exquisite manner," Kim says. "If we hadn't come across the paper that led us to the hydrogen-exchange, we would not have been able to discover that."
More information: Colin Y. Kim et al, Emergence of a proton exchange-based isomerization and lactonization mechanism in the plant coumarin synthase COSY, Nature Communications (2023). DOI: 10.1038/s41467-023-36299-1
Journal information: Nature Communications
Provided by Whitehead Institute for Biomedical Research