This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Bacteria rely on cooperation and evolution to defend against predatory protists, finds study
Eating and being eaten is a normal process in nature. These predator–prey dynamics help to stabilize ecosystems. It ensures that individual species do not become too abundant, controls their populations, and prevents damage caused by overpopulation (e.g. browsing by deer in the forest or damage to crops by caterpillars).
But how is it that the predators do not simply eat away all the prey, thus breaking down the system? A research team from the Helmholtz Centre for Environmental Research (UFZ) together with scientists from the Technical University (TU) of Dresden and the University of Potsdam has investigated this using bacteria and protists that live in bodies of water and discovered something astonishing.
According to an article recently published in the ISME Journal, bacteria defend themselves against predatory protists with cooperative behavior and evolution.
In a lake or river, between one and 10 million bacteria live in just 1 ml of water. Such a high density is necessary because bacteria permanently break down organic compounds and pollutants and thus purify the water. However, if there are too many bacteria, this can lead to the spread of pathogens. Preventing this requires predators: microscopic protists of which there are usually between a few hundred and a few thousand individuals in 1 ml of water.
They constantly eat bacteria and thus ensure that the bacteria fulfill their cleaning function but do not become too abundant. Using the bacterium Pseudomonas putida and the bacterivorous protist Poteriospumella lacustris, the research team investigated the role of the various defense strategies of the bacteria and how the formation of feeding resistance is related to the dynamics of ecological systems.
Cooperative behavior helps—but only in the short term
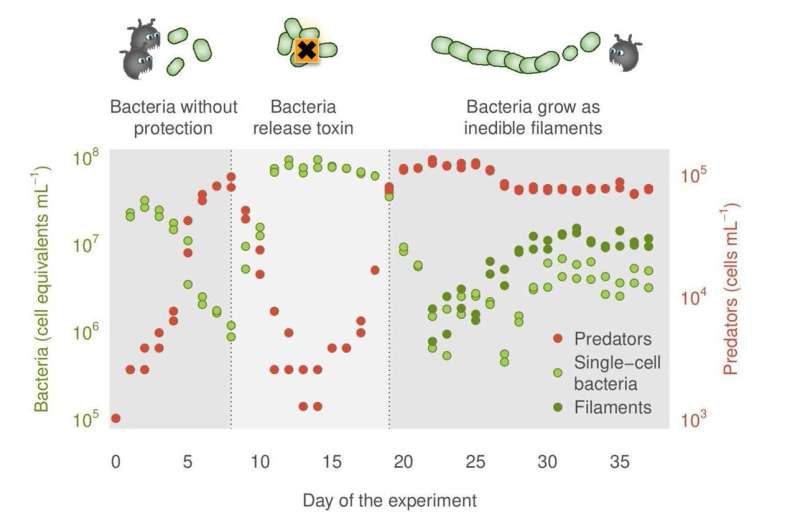
In the five-week laboratory experiment, the scientists found that, as expected, the predatory protists first multiplied in the bacterial culture for a week and reduced the number of bacteria. However, the population of protists quickly collapsed in the second week because the bacteria produced a toxin that strongly inhibits the reproduction of these predators.
"Such a chemical defense is successful only if a relatively large number of bacteria join in and release a corresponding amount of toxin into the water," says Dr. Magali de la Cruz Barron, lead author and hydrobiologist at the UFZ and TU Dresden.
This cooperative behavior protects the entire population—at least for a short time. But after a few days, the bacteria no longer secreted toxin, and the predators recovered by the end of the third week. It is not possible to say exactly why this is. One common explanation for such a phenomenon is that too many "cheaters" form.
These are in this case bacteria that do not form toxins themselves but which benefit from them in the group because they do not have to expend any effort of their own to protect themselves. "But we could show with the help of mathematical simulations that cheaters are not necessary to explain such patterns if alternative defense strategies evolve," explains Magali de la Cruz Barron.
Individual defense lasts a long time and stabilizes the population densities
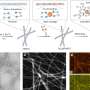
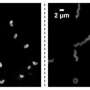
And indeed, the research team discovered a second defense mechanism that the bacteria developed from the third week onwards. Most bacteria formed filaments (i.e. threads with cells arranged in chains). These made the bacteria 10 to 100 times larger and much bulkier so that many of them could no longer be eaten by the protists. This individual behavior was successful.
The bacterial density stabilized by the end of the fifth week. However, there were still sufficient numbers of bacteria that could be eaten because in order to reproduce, the bacteria had to keep forming small units that served as food for the predators. This also allowed the protists to establish a stable population density. Unlike toxin formation, the individual defense of the bacteria was not reversible.
"By sequencing the bacterial genome, we have proven that the formation of the filaments was indeed accompanied by a change in the genetic material. Evolution has thus taken place. Not over millions of years but rather within only a few days," says Prof. Dr. Markus Weitere, co-author and head of the UFZ Department of River Ecology. This observation is not entirely new. It is known that evolution can take place in relatively short periods of time, especially in fast-growing organisms such as bacteria.
"But what is remarkable is that this mutation did not happen just once. The experiments were often repeated, and these adjustments were always made," says Weitere. Even though the change in the genome probably occurs by chance, it leads to a reproducible adaptation pattern in the bacteria.
With this experiment, the research team showed how the formation of defense strategies affects the dynamics of predator–prey systems and how important this defense is for stabilizing populations. It also became clear that it makes sense for the prey species not to rely on just one strategy.
"Depending on the situation, one of several strategies can be successful. In our experiment, it was the quick cooperative behavior that led to the initial success. In the end, it was the more cumbersome individual defense through evolution that led to a permanent defense," says Weitere. Thus, individual protection prevails—even if the initial cooperative defense was definitely beneficial for the community.
More information: Magali de la Cruz Barron et al, Shifts from cooperative to individual-based predation defense determine microbial predator-prey dynamics, ISME Journal (2023). DOI: 10.1038/s41396-023-01381-5. www.nature.com/articles/s41396-023-01381-5
Journal information: ISME Journal
Provided by Helmholtz Association of German Research Centres