This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Study lays out chirality-flipping theory
Chemists can make a career out of controlling whether certain molecules are generated as a lefty or a righty.
Molecules don't literally have hands, but scientists often refer to them in this way when looking at molecules that are mirror images of each other and therefore are not superimposable. And whether a molecule is a lefty or a righty directly affects how they behave and their use in everything from drug design to flavoring foods.
A Florida State University research team led by Associate Professor of Chemistry Ken Hanson previously found a way to turn "left-handed" molecules into "right-handed" ones by using light to induce a proton transfer and the transformation into a different isomer. Now, Hanson and his fellow FSU Professor of Chemistry Eugene DePrince are harnessing the power of math and computers to predict what would happen if you performed that same process in a gap between closely spaced mirrors.
The results, published in the Journal of Physical Chemistry A, suggests that scientists could use these cavities to control the degree of the change in handedness, also referred to as chirality.
"Although this paper is entirely theoretical, it can guide experimentalists to rethink how to control the handedness of reactions," Hanson said.
The understanding and control of chirality, the property of asymmetry in molecules, is of particular importance for pharmaceuticals, fragrances, food additives and more. Using the wrong-handed molecule can have severe consequences.
In the 1950s, a sedative called thalidomide was sold in Europe as a treatment for morning sickness. It was later discovered that while the right-handed molecule for thalidomide was effective, the left-handed one caused sever birth defects in thousands of children whose mothers took the drug.
Because of this and other cases, pharmaceutical companies and the FDA approval process are hyper-focused on the left-hand versus right-hand issue in their drug screening.
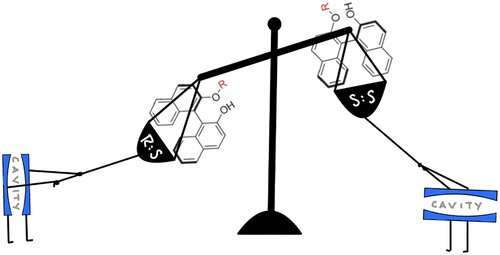
In a previous study, Hanson used an organic compound called BINOL and attached a left-handed amino acid to it. Then, the team shone light directly on the compound. This reaction changed a 50-50 mixture of left and right-handed BINOL into a predominantly right-handed mixture.
DePrince was intrigued by the study and suggested they perform simulations that could offer predictions on what would happen if this reaction occurred between reflective surfaces.
"Many studies of cavity-mediated chemistry are driven by theory," DePrince said. "Engineering a cavity that is small enough to achieve the effect we see is difficult, but our simulations allow us to at least predict what can happen."
In addition to showing that this process could change the degree of handedness, it also demonstrated that changing how the molecule is oriented between the mirrors can influence which handedness, right or left, the chemical reaction produces.
"This latter point is particularly remarkable because usually you have to use some sort of chiral directing auxiliary, or a particularly handed directing agent, to impact the handedness of your reaction," Hanson said. "Here the mirrors do all the work."
More information: Nam Vu et al, Enhanced Diastereocontrol via Strong Light–Matter Interactions in an Optical Cavity, The Journal of Physical Chemistry A (2022). DOI: 10.1021/acs.jpca.2c07134
Journal information: Journal of Physical Chemistry A
Provided by Florida State University