This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Elucidation of electrolyte decomposition behavior in all-solid-state lithium-sulfur batteries
A research group of the Department of Electrical and Electronic Information Engineering at Toyohashi University of Technology—consisting of Hirotada Gamo, a doctoral course student; Kazuhiro Hikima, assistant professor; and Atsunori Matsuda, professor—has elucidated the decomposition behavior of electrolytes in the cathode composites of all-solid-state lithium-sulfur batteries (ASSLSB).
It was found that the sulfide solid electrolytes in the ASSLSB's cathode composites were converted into thiophosphates with long-chain cross-linked sulfur through the charging and discharging cycle. These decomposition products govern the overall battery performance of ASSLSBs. The study was published in the journal Chemistry of Materials.
Currently, electric vehicle (EV) market is expanding and thus require the development of better onboard storage batteries. ASSLSBs are expected to become next-generation high-energy-density batteries owing to the use of high-capacity cathode active materials, such as sulfur (S) and lithium sulfide (Li2S).
In order to ensure that ASSLSBs offer optimal performance, robust ion and electron conduction pathways must be established within the cathode composites, which are comprised of cathode active materials with sulfide solid electrolytes and a conductive agent (carbon material).
However, battery performance is presently limited due to insufficient ionic conduction pathways provided by the sulfide solid electrolytes in the cathode composite. Also, sulfide solid electrolytes cause the oxidative decomposition within the ASSLSB's operating voltage. Excessive decomposition of solid electrolytes results in the loss of ionic conduction pathways within the cathodes, and thus to degraded capacity.
It is therefore important to understand the decomposition behavior of sulfide solid electrolytes in cathode composites in order to improve ASSLSB performance. In according to previous reports, sulfide solid electrolytes are converted into redox-active decomposition products after the oxidation decomposition, which show reversible redox reactions. However, how sulfide solid electrolytes decompose in ASSLSBs remains unclear.
Accordingly, the research team used planetary ball milling to mix Li2S as the cathode active material, Li3PS4 as the sulfide solid electrolyte, and carbon material to fabricate the cathode composite; on which they then conducted electrochemical measurements and Raman spectroscopy, and analyzed how the electrolyte in the cathode composite of the ASSLSBs decomposed during charging and discharging.
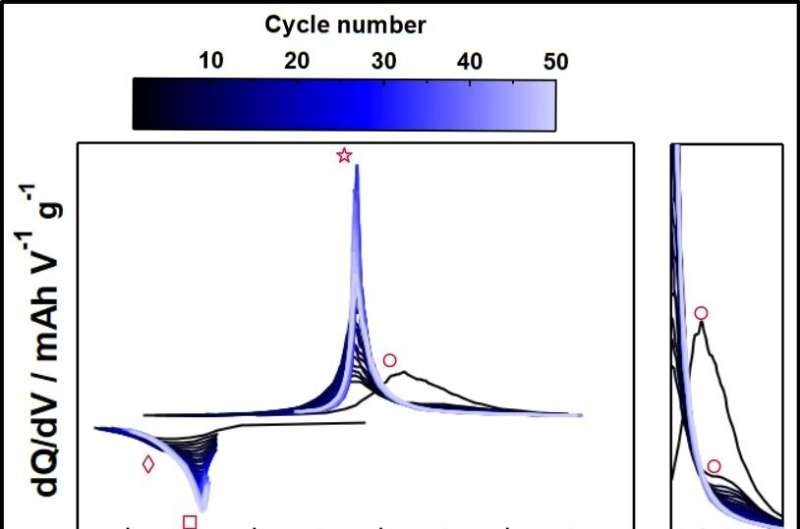
Basic electrochemical measurements revealed that redox reactions in ASSLSBs include the oxidative decomposition of Li3PS4 and redox reactions of its decomposition products, as well as redox reactions of the cathode active materials.
After prolonged repetition of these cycles, the cathode active materials lost their electrochemical activity, and only the redox reactions of Li3PS4 decomposition products remained evident. This demonstrates that the long-term performance of ASSLSBs is governed by the electrochemical redox activity of Li3PS4 decomposition products.
Raman spectroscopy on the cathode composite after 20 cycles revealed a peak derived from a S-S bond. These experiments demonstrated that the cathode active materials react with the sulfide solid electrolytes in ASSLSBs during the charging and discharging cycle to form thiophosphates with long-chain cross-linked sulfur.
More information: Hirotada Gamo et al, Understanding Decomposition of Electrolytes in All-Solid-State Lithium–Sulfur Batteries, Chemistry of Materials (2022). DOI: 10.1021/acs.chemmater.2c02926
Provided by Toyohashi University of Technology