More than 52 million birds in the US have been affected by an outbreak of avian influenza
A form of highly pathogenic avian influenza (HPAI) has been causing widespread disruption and illness in birds since first being detected roughly a year ago. Last March, a bald eagle found dead in Pennsylvania's Chester County became the first confirmed case in the state.
Since then, through the Wildlife Futures Program and the Pennsylvania Animal Diagnostic Laboratory System (PADLS), Penn's School of Veterinary Medicine has ramped up its HPAI-related diagnostics work, supporting agencies including the Pennsylvania Game Commission and the U.S. Department of Agriculture in monitoring for the disease in both wild and domestic birds.
The outbreak, caused by the H5N1 HPAI strain, has surpassed a milestone: more than 50.5 million birds affected. The figure outstrips that of a 2014-15 HPAI outbreak, which at the time was considered "the single biggest animal health emergency in U.S. history based on the number of birds involved," says Lisa Murphy, who heads the PADLS laboratory at Penn Vet's New Bolton Center campus.
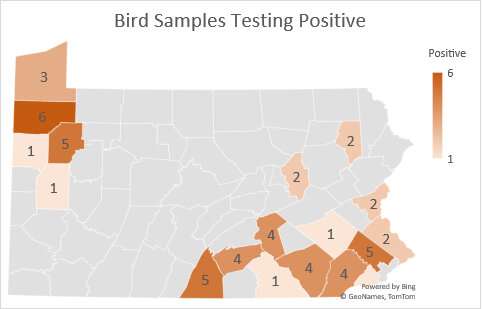
While avian influenza viruses typically peter out during the summer when wild birds end their migrations, this outbreak came roaring back in the fall and is now in 46 states, with Pennsylvania experiencing particularly severe losses of more than 4 million birds. Why remains an open question, but new lines of research by Penn Vet researchers including Eman Anis, a microbiologist, and Louise Moncla, who joined the faculty in September, aim to understand—at the genetic level—the reasons this particular virus is causing so much harm.
Tracking a virus through tests
Even when there is not an active disease outbreak, PADLS regularly performs avian influenza tests as part of statewide surveillance efforts. "We are available every weekday and are on call over the weekends," says Anis, an assistant professor of microbiology.
Last winter, even before this viral strain arrived in Pennsylvania, the lab began stocking supplies, enough to run at least 500 samples a day. "And while typically we only test samples from domestic poultry, we have been receiving samples of wild birds through our Wildlife Futures Program," Murphy says, including the bald eagle which marked the state's first confirmed case of HPAI. Samples that are suspected to be HPAI-positive are sent for confirmatory testing to the National Veterinary Services Laboratory.
Testing at PADLS takes place in a secure Biosafety Level 2 lab at New Bolton Center and involves a PCR test, short for polymerase chain reaction, that looks for particular sequences in the genome that are characteristic of certain viruses. If that test reveals the presence of influenza A virus, the group to which HPAIs belongs, then further PCR testing can determine the subtype.
Viruses that are found to possess a hemagglutinin (H) protein 5 or 7 undergo additional testing to determine whether they are high- or low-pathogenicity. This distinction has to do with how the virus's hemagglutinin protein attaches to host cells. Mutations that give the virus the ability to bind to more types of host cells and spread to and infect cells more easily are likely to cause more severe disease.
While low-pathogenicity influenza viruses may cause mild symptoms that could be missed, high-pathogenicity viruses can affect multiple organ systems. "Some affected birds have neurological signs, so they may have difficulty flying or appear disoriented," Anis says.
When a so-called "high-path" virus is found in a commercial flock, typically the whole farm must cull its birds. This happened to three operators in Pennsylvania's Lehigh Valley in the lead-up to Thanksgiving, the farms sacrificing more than 60,000 birds "at the worst possible time," Murphy says.
How viruses evolve, spread, and wreak havoc
Penn Vet's PADLS lab recently sought and received permission from the U.S. Department of Agriculture and biosafety regulators at Penn to keep some non-infectious genetic material from the samples they test for HPAI. Researchers including Anis and Moncla plan to soon begin analyzing this RNA to understand the virus's evolution and ability to cross species.
Moncla will rely in part on digital tools she curated to help visualize these patterns and transmission routes for a variety of pathogens, including the avian H5N1 virus. An open-source platform she began working on during her postdoctoral fellowship called Nextstrain enables users to monitor what is known about the relationship between detected pathogens. The data there inform a phylogenetic tree of H5N1—in other words, the family tree of the virus—that indicates the strain currently circulating in the U.S. originated in China, spread to Southeast Asia and then Europe, before moving into North America.
What isn't as clear is why the virus is behaving so differently from previous avian influenza strains. The strain, like most H5N1 HPAIs, doesn't typically target people; just two human cases have been confirmed, both of which were in individuals who work closely with birds. Yet while certain bird flu viruses are somewhat limited in the species they affect, this H5N1 virus appears to be less choosy.
Infections have been found in a broad range of birds as well as some mammals, including fox, bear, and even seals. Scientists have also seen an extended transmission of disease within wild bird populations that carried beyond the spring migration season and is ticking up again with the fall migration.
"Traditionally, it was thought that wild waterfowl, like ducks and geese, were the primary concern when it comes to transmission," says Murphy. "But with this one, we've been seeing a lot of problems with raptors, birds of prey like bald eagles and vultures. With vultures in particular they roost in large groups, so you can imagine the virus sweeping quickly through a population."
The strain's apparent ability to affect wild birds and move between wild and domestic populations, says Moncla, may be traceable to particular genetic variations that enable the hemagglutinin protein to bind a wider-than-normal range of host species' cells, or there could be other factors at play. Just which ones are responsible are what she hopes to uncover by sequencing the virus in her lab.
"There's currently no public data on avian influenza genomics from Pennsylvania despite the fact that there's a big outbreak here," Moncla says. "My goal would be to find actionable information: How is this virus moving between domestic and wild birds in the state? Are there particular species that are acting as sources of new genetic diversity? I think we have the opportunity to be on the forefront of watching this virus as it becomes endemic in North America."
Provided by University of Pennsylvania