New method of reducing carbon dioxide could be a golden solution to pollution
![A gold-based hybrid material (Au@CB[6]) is modified by CB[6] in order to efficiently convert CO2 to CO. Credit: Nano Research, Tsinghua University Press New method of reducing carbon dioxide could be a golden solution to pollution](https://scx1.b-cdn.net/csz/news/800a/2022/new-method-of-reducing.jpg)
Reducing carbon dioxide (CO2) to carbon monoxide (CO) through an electrochemical reaction holds strong potential for removing CO2 from the atmosphere to reduce pollution and for producing alternative energy with carbon monoxide as an ingredient.
However, the current catalysts used in electrochemical carbon dioxide reduction reaction (CO2RR) are not efficient or selective enough to make CO2RR a practical solution. Now, a team of researchers from Fujian Institute of Research on the Structure of Matter of Chinese Academy of Sciences has developed a gold-based hybrid material by modifying gold nanoparticles with a macrocyclic compound called cucurbit[6]uril (CB[6]) that allows for more efficient CO2RR than previously possible.
The results were published in Nano Research.
"With this work, we hoped to solve the problem of environmental pollution and energy shortage through electrochemical conversion of carbon dioxide to value-added products," said corresponding author Minna Cao of the State Key Laboratory of Structural Chemistry, Fujian Institute of Research on the Structure of Matter, Chinese Academy of Sciences, and of the University of Chinese Academy of Sciences.
"In order to enhance the local CO2 concentration on the catalysts' surface, we utilize macromolecule cucurbit[n]uril to functionalize gold surface, which is the distinguishing feature of our work from those that has been done before."
According to the researchers, gold is known to be highly active in converting CO2 to CO relative to other catalysts. However, the binding energy of both CO2 and CO to the gold catalyst surface is positively correlated, which clashes with the need in CO2RR for CO2 adsorption and CO desorption, since the CO desorption does not occur due to the positive correlation of its binding energy to the catalyst.
The researchers created a controlled synthesis of nanoparticles by modifying CB[6]. CB[6] has negatively charged portals and positively charged surface, which helps contribute to the result of having the electronic interaction between CB[6] and metal regulate the catalytic performance.
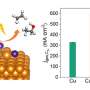
The researchers verified both the morphology and surface structure of the nanoparticles through transmission electron microscopy. The gold-based hybrid material (Au@CB[6]) was proven to enhance CO2RR catalytic activity.
"We have proved the interaction between cucurbit[6]uril and CO2 through operando electrochemical measurement and density functional theory calculations," Cao said.
According to the researchers, multiple factors contribute to the improved catalytic performance. First, the CB[6] can increase the local CO2 concentration near the metal surface by gathering CO2, which means that the Au@CB[6] has tunable, or adjustable, CO2 enrichment. Additionally, the modification of CB[6] allow for improved CO2RR by breaking the previously mentioned scaling relations of the binding affinity between catalyst surface and CO2/CO.
Also, one reason that CO2RR was limited in efficiency with gold surface catalysts previously is that CO2 has low solubility in aqueous electrolytes, an issue that the researchers solved by using the highly specific binding force of macrocycle to selectively adsorb certain species to regular the electrocatalytic reaction.
"The results showed that CB[6] can gather CO2 and lead the increased local CO2 concentration near the metal interface, as well as promote CO desorption, which are the dominating reasons for enhanced CO2RR performance," Cao said. "Using the rigid macrocycle cucurbit[n]uril to modify the catalysts' surface is a promising pathway to enhance the electrocatalytic performance."
The researchers plan to continue to modify the catalyst in order to further improve the efficiency of the CO2RR.
"In the next step, we hope to adjust the shape and size of the gold catalyst in the presence of cucurbit[n]uril to further promote the catalytic performance toward electrochemical reduction of carbon dioxide to value-added products," Cao said.
More information: Huimin Wang et al, Tunable CO2 enrichment on functionalized Au surface for enhanced CO2 electroreduction, Nano Research (2022). DOI: 10.1007/s12274-022-5159-8
Journal information: Nano Research
Provided by Tsinghua University Press