Enhanced single-nanoparticle collisions for the hydrogen evolution reaction in a confined microchannel
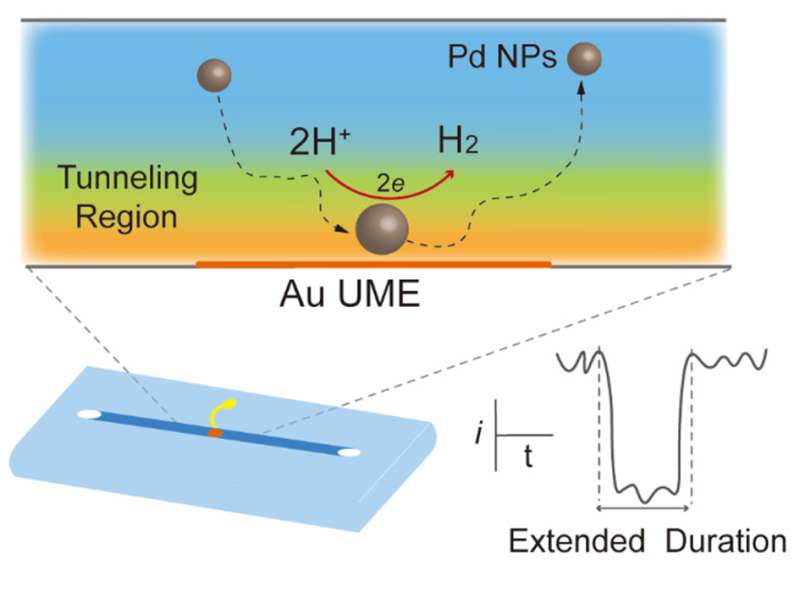
Single-nanoparticle (NP) collisions have enabled the measurement of electrocatalytic reactions on single NPs by the nanoconfinement-amplified current. Recently, it has been demonstrated that the electrocatalytic activity of single NPs is improved by stochastic collision electrochemistry, because the good dispersion alleviates the aggregation, deactivation, and detachment of nano-electrocatalysts adhered on the electrode surface. However, there is still a pressing need to further increase the electrocatalytic performance of single NPs.
At the most fundamental level, the electrochemical reaction can only be triggered when single NPs reach the tunneling distance. Therefore, increasing the collision period of single NPs in the tunneling region should contribute to the improvement in the electrocatalytic efficiency.
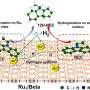
Recently, a research team led by Prof. Yi-Tao Long from Nanjing University, China report the use of a confined microchannel to control the dynamic motion of single Pd NPs, with the aim of building a platform to increase the collision period of single Pd NPs in the tunneling region. Single Pd NP collisions serve as a model system that could be used to easily investigate high-performance catalysts for the hydrogen evolution reaction (HER).
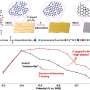
They show that the integrated charge associated with the HER in the confined channel reaches twice as high as that based on conventional single-NP collisions due to the enhanced hydrodynamic trapping effect. The results were published in Chinese Journal of Catalysis.
The microchannel is first cast in PDMS, then attached to a glass slide supporting an Au UME, following performed stochastic collision electrochemical measurements. It is worth noting that the collision duration of Pd NPs using the confined microchannel is longer than that by using the conventional UME. Near-wall hindered diffusion would be significant when NPs are dispersed in a confined microchannel, indicating that the diffusion of single NPs is hindered by additional hydrodynamic adsorption.
Consequently, near-wall hindered diffusion effect that could confine dynamic single Pd NPs in the tunneling region at where the HER possibly occurs. Further, the reaction time during a collision is extended.
The single Pd NP collisions with the microchannel-based Au UME were performed at different applied overpotentials to further demonstrated the effect of an extended collision duration on the HER kinetics. According to the information on the electrochemical characteristics of Pd NPs for HER in the ensemble system, it is proved that the time-resolved current signals is generated by charge displacement at the double layer of the Au UME due to the dynamic motion of single Pd NPs, hydrogen under-potential desorption, and HER on single Pd NPs, respectively.
More information: Si-Min Lu et al, Enhanced single-nanoparticle collisions for the hydrogen evolution reaction in a confined microchannel, Chinese Journal of Catalysis (2022). DOI: 10.1016/S1872-2067(21)64034-2
Provided by Chinese Academy of Sciences