New study finds that CRISPR/Cas9 leads to unexpected genomic changes

The widely used gene scissor (CRISPR/Cas) can modify the genetic content in cells to study the molecular roles of genes and has gained great clinical relevance in gene therapy to treat genetic diseases. A new study performed by Claudia Kutter's research group at the Department of Microbiology, Cell, and Tumor Biology at Karolinska Institutet, found that the gene scissor leads to unexpected genomic changes.
"We found that CRISPR/Cas9 causes unpredictable on-target genomic effects that impacted cancer cell growth. Instead of slowing down, cancer cells grew faster. Given that CRISPR/Cas genome engineering has become a common tool in research and clinical applications, we believe that our novel insight, as well as our innovative and robust approach, will be useful in evaluating genomic changes," says corresponding author Claudia Kutter, principal researcher at the Department of Microbiology, Cell and Tumor Biology, Karolinska Institutet.
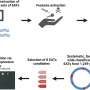
The research team developed a new approach to decipher the genomic changes at high resolution and to uncover varying genomic effects at the targeted locus.
"To explain the surprising phenotype of cells, we combined the latest technological approaches. First, we specifically enriched our targeted genetic sequence in microdroplets (Xdrop). Second, we read the genetic sequence by using nanopore long-read sequencing technology (ONT-LRS). Finally, we developed a sophisticated computational pipeline that deciphered the exact changes in the genomic sequence," says first author Keyi Geng, a Ph.D. student in Claudia Kutter's research group.
The paper is published in Genome Research. The research team is now investigating other CRISPR/Cas effects in human cells, with a special focus on cancer cells that have escaped cancer treatments.
"This helps us understand how our genetic material is repaired if damaged. Although CRISPR/Cas is very powerful, we need to find better ways to control CRISPR/Cas activity in human cells. Making CRISPR/Cas safer for patient treatments is therefore a major effort in my group," Claudia Kutter says.
More information: Keyi Geng et al, Target-enriched nanopore sequencing and de novo assembly reveals co-occurrences of complex on-target genomic rearrangements induced by CRISPR-Cas9 in human cells, Genome Research (2022). DOI: 10.1101/gr.276901.122
Journal information: Genome Research
Provided by Karolinska Institutet