New strategies to accelerate application of lithium-rich Mn-based cathode
Lithium-rich Mn-based oxide (LRMO) is one of the next generation cathode materials for lithium-ion batteries (LIBs), which is expected to exceed high energy density of 550 Wh kg-1.
However, the redox reaction of anionic oxygen (O2-) is sluggish and not stable, leading to poor rate capability and cycling performance of LIBs, especially in solid-state batteries.
Recently, a research team led by Prof. Cui Guanglei from the Qingdao Institute of Bioenergy and Bioprocess Technology (QIBEBT), Chinese Academy of Sciences (CAS), has provided new insights in revealing heterogeneous Li+ transport dynamics and regulating anion oxygen stability of LRMO cathode materials.
LRMO materials have better prospects for application in solid-state batteries than commercial cathode materials due to their high specific capacity and low cost. "It is an important premise to solve the key scientific problems of LRMO by clarifying the micro-mechanism of degradation and developing innovative material preparation technology," said Prof. Cui from QIBEBT.
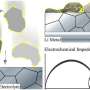
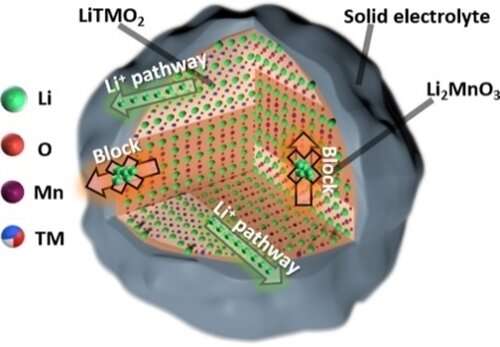
The team for the first time observed the heterogeneous Li+ transport behavior of LRMO in sulfide solid-state batteries using the scanning transmission electron microscopy technique of in-situ differential phase contrast imaging (STEM-DPC). They found that the nano-scale two-phase separation (NCM111 phase and Li2MnO3 phase) in LRMO was the decisive factor for the Li+ transport heterogeneity in the bulk phase and interface of cathode and solid electrolyte, which severely restrains the capacity contribution of lithium-rich Li2MnO3.
Their study was published in Angewandte Chemie International Edition.
"We investigated the 'structure-activity relationship' between microstructure, Li+ transport kinetics and electrochemical performance of LRMO, and clarified the micro-mechanism of performance attenuation for LRMO cathode in solid-state batteries," said Ma Jun, associate professor at QIBEBT. This study further manifests the importance of accurately optimizing crystal structure and enhancing the Li+ transport kinetics at cathode/electrolyte interface.
In another study published in Advanced Energy Materials, the researchers proposed a new material preparation technology of swing-like non-isothermal sintering (SNS), which stabilized the lattice oxygen in bulk phase of LRMO and reduced the generation of unsteady O2p holes.
Compared with the traditional constant-temperature sintering (CTS) technology, the electrochemical performance of the cathode prepared by SNS technology, such as specific discharge capacity and cycle stability, have been improved.
In addition, the feasibility of SNS technology has also been verified in the cobalt-free lithium-rich manganese-based cathode material system (Li1.2Mn0.6Ni0.2O2).
"This study will provide guidance for stabilizing the anion oxygen structure and elevating the integral electrochemical properties of LRMO materials," said Zhang Yuhan, the first author of the study.
"The above works establish a solid foundation for exploiting the LRMO-based solid-state batteries with high energy density and high safety," said Prof. Cui.
More information: Bowen Liu et al, Direct Observation of Li‐Ion Transport Heterogeneity Induced by Nanoscale Phase Separation in Li‐rich Cathodes of Solid‐State Batteries, Angewandte Chemie International Edition (2022). DOI: 10.1002/anie.202209626
Yu‐Han Zhang et al, Stabilization of Lattice Oxygen in Li‐Rich Mn‐Based Oxides via Swing‐like Non‐Isothermal Sintering, Advanced Energy Materials (2022). DOI: 10.1002/aenm.202202341
Journal information: Angewandte Chemie International Edition , Advanced Energy Materials
Provided by Chinese Academy of Sciences