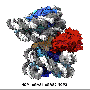Researchers identify Ku proteins as new co-sensors of cyclic GMP-AMP synthase
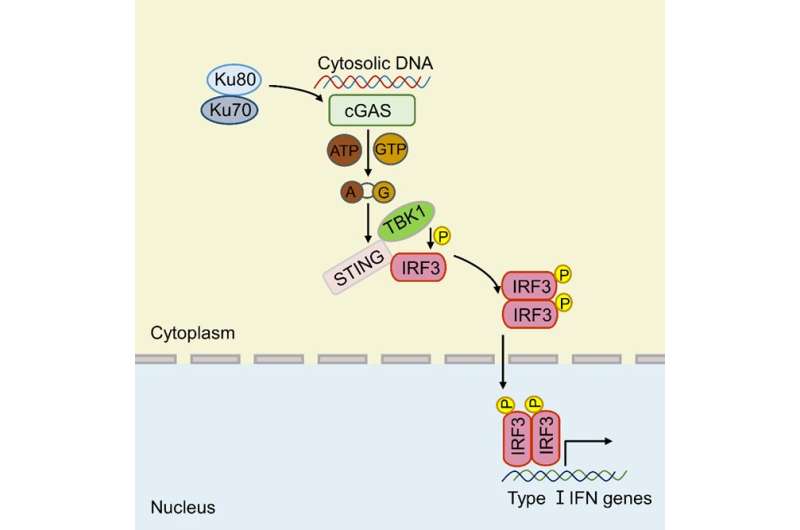
The initiation of the innate immune responses relies on the detection of microbes by multiple pattern recognition receptors. Among these receptors, cyclic GMP-AMP synthase (cGAS) has been identified as a major and universal cytosolic DNA sensor that is independent of specificity of DNA sequence and/or cell type. However, previous studies have suggested that the binding affinity of cGAS to DNA is relatively low, how the low binding affinity between cGAS and DNA still efficiently mediates the innate immune responses remains largely unknown.
In a recent study published in Cell Reports, researchers from the Institute of Zoology of the Chinese Academy of Sciences and Yunnan University revealed that Ku proteins act as co-sensors of cGAS to regulate cGAS-STING signaling pathway.
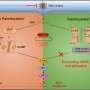
According to the researchers, upon DNA binding, cGAS undergoes aggregation that leads to the activation of cGAS. Activated cGAS catalyzes the synthesis of the second messenger cyclic GMP-AMP (cGAMP) from ATP and GTP. cGAMP binds to and activates the adaptor protein STING, and STING activates downstream signaling to produce type I interferons and proinflammatory cytokines.
Of note, the direct binding affinity between cGAS and DNA is relatively low; this raises a possibility that co-sensors may act in concert with cGAS to mediate efficient antiviral signaling.
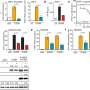
In this study, the researchers identified Ku proteins as positive modulators of the cGAS-mediated signaling pathway. They demonstrated that Ku proteins directly interacted with cGAS, promoted the DNA binding affinity of cGAS, augmented its condensation, and enhanced its enzyme activity to produce more cGAMP, thereby facilitating the activation of cGAS-STING signaling pathway.
This work provides insights into understanding the effective regulatory mechanism of cGAS-mediated innate immune signaling.
More information: Xinyue Tao et al, Ku proteins promote DNA binding and condensation of cyclic GMP-AMP synthase, Cell Reports (2022). DOI: 10.1016/j.celrep.2022.111310
Journal information: Cell Reports
Provided by Chinese Academy of Sciences