Foreign vs. own DNA: How an innate immune sensor tells friend from foe
How do molecules involved in activating our immune system discriminate between our own DNA and foreign pathogens? Researchers from the Thomä group, in collaboration with the EPFL, deciphered the structural and functional basis of a DNA-sensing molecule when it comes in contact with the cell's own DNA, providing crucial insights into the recognition of self vs. non-self DNA.
DNA within our cells is compacted and stored in the nucleus in the form of chromatin (DNA wraped around histone proteins, forming nucleosomes, the basic unit of chromatin). DNA found outside the nucleus, in the cytoplasm, is an important signal that triggers immune responses indicating the presence of an intracellular pathogen or a potentially cancerous cell. DNA sensing is carried out by cGAS, an enzyme responsible for recognizing and binding naked DNA. When activated, cGAS synthesizes cyclic GMP-AMP, which in turn initiates the body's so-called "innate" immune system—the first-line-of-defense part of our immune system.
Until now, cGAS was thought to function predominantly in the cytoplasm, detecting foreign, non-self, DNA such as viruses. But recent studies suggested that cGAS is also present inside the nucleus. This was puzzling given the possibility that the enzyme is activated by its own DNA triggering an unwanted inflammatory response against its own DNA. Intrigued by this observation, researchers from the Thomä group used structural biology as a discovery tool and found that cGAS is present in the nucleus in an inactive state. They teamed up with the Ablasser lab at the EPFL to decipher the mechanism of cGAS inactivation by chromatin in cells.
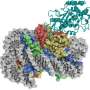
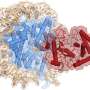
Taking advantage of the capability of the Thomä lab in cryo-electron microscopy (cryo-EM), the researchers derived the structure of cGAS bound to a nucleosome. They found that cGAS directly engages the histone proteins of nucleosomes. Once bound to the nucleosome, cGAS is "trapped" in a state in which it is unable to engage or sense naked DNA. It is then also unable to synthesize GMP-AMP and remains inactivated. cGAS, when present in the nucleus of healthy cells, is thus inactivated by chromatin, and does not participate in innate immune signaling in response to its own DNA.
Ganesh Pathare, a postdoc in the Thomä lab and one of the first authors of the study, comments: "The cGAS-nucleosomes structures provide the structural and functional basis for cGAS inhibition by chromatin. cGAS is an important protein for the innate immune response in the cell, required for the fight against viruses but also for detecting transformed or cancerous cells. cGAS activity is also often misguided in autoimmune diseases. Our study provides crucial insights into cGAS regulation and the mechanism of self DNA vs. non-self DNA recognition. This creates exciting opportunities for future therapeutic intervention in a wide range of diseases".
This study was published in the 26 November 2020 issue of Nature.
More information: Ganesh R. Pathare et al. Structural mechanism of cGAS inhibition by the nucleosome, Nature (2020). DOI: 10.1038/s41586-020-2750-6
Journal information: Nature