Study unlocks key to success of drug-resistant bacteria
Researchers have discovered how Gram-negative bacteria—which cause drug-resistant pneumonia, bloodstream infections, and surgical site infections in hospitalized patients—finish building a crucial component of their outer membrane that shields these pathogens from attacks by the immune system and antibiotics. The new findings could accelerate the development of novel drugs to counteract these potentially deadly bacteria, the source of countless infections in health care settings worldwide.
The study was published online April 6 in Nature.
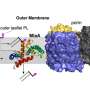
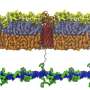
"Previously, we knew that Gram-negative bacteria construct their outer membrane with two main, non-protein components—lipids and sugars—which together form an impermeable barrier. The missing link was how this lipopolysaccharide component comes together," says Filippo Mancia, Ph.D., a co-leader of the study and professor of physiology & cellular biophysics at Columbia University Vagelos College of Physicians and Surgeons.
Using cutting-edge single-particle cryo-electron microscopy, Mancia and colleagues were able to determine the structures of the enzyme that links together the lipids and sugars (called an O-antigen ligase), in two different functional configurations. Then, combining genetic, biochemical, and molecular dynamics experiments, the team learned how the enzyme positions the lipids and sugars so that they can combine to form the protective membrane.
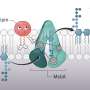
The lipopolysaccharide component of the outer membrane is critical to the survival of Gram-negative bacteria. "If you could block its assembly, then you would make the bacteria more sensitive to antibiotics and more vulnerable to the immune system," Mancia says.
Construction of this membrane is an ongoing process, beginning when Gram-negative bacteria are first formed and continuing as the membrane naturally degrades and requires repair. "This means that we would have many opportunities to disrupt the membrane, not just at one stage of the bacteria's life cycle," Mancia says.
After revealing the structure of the enzyme that performs the last and critical step in assembling lipopolysaccharide barriers in drug-resistant bacteria, the researchers may begin to custom-design drugs that inhibit the biosynthesis of this protective membrane.
The paper is titled "Structural basis of lipopolysaccharide maturation by the O-antigen ligase."
More information: Khuram U. Ashraf et al, Structural basis of lipopolysaccharide maturation by the O-antigen ligase, Nature (2022). DOI: 10.1038/s41586-022-04555-x
Journal information: Nature
Provided by Columbia University