Researchers find 'genetic baggage' accumulates in the genomes of aging mutant animals

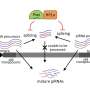
You are probably familiar with the term that some people carry "a lot of extra baggage." Usually that term refers to that person's emotional history, but in genetics and our genomes, "extra baggage" can also describe the transposons lurking in our genomes, a historical record of our genomes surviving traumatic invasions during evolution. Transposons are repetitive DNA sequences that have the capability to move (transpose) from one location to another in the genome (an organism's complete set of genetic instructions) and are considered important invaders of our genomes during evolution.
"Typically, when we are young and healthy, our genomes do a good job handling all these transposons, keeping them stored away. But what if during aging or in unhealthy mutants, how does one handle all this 'extra baggage' in our genomes?" a question asked by corresponding author Nelson Lau, Ph.D., associate professor of biochemistry at BUSM and Director of the BU Genome Science Institute.
In a new study in the journal of PLoS Genetics, researchers from Boston University School of Medicine (BUSM) tested this question of how these mobile genetic elements can accumulate in the genomes of normal and mutant animals during a single life span. Their study used the model organism of fruit flies which have 12 percent of their genome made up of transposons and are a good proxy for humans, where transposons make up over 40 percent of our DNA.
According to the researchers, typically, young animals store away transposons neatly, so they remain organized and quiet. However, some researchers are now seeing transposons becoming activated during animal aging, when the natural processes to silence transposons decline with age. "Slowing the ravages of aging continues to be an important goal of biomedical research, and this study aimed to determine if transposons activated during aging can move and accumulate in older genomes," says Lau.
The Lau lab team used whole genome sequencing and bioinformatics to show that normal older fruit flies can keep transposons from accumulating in genomic DNA even though the transposon RNAs were still elevated during aging. However, sicker animals with mutations affecting RNA interference pathways appeared to pile up many more of these transposon copies in the genome during aging.
To see if this condition could be treated, the investigators used genetic tricks to improve the RNA interference pathways in flies, this helped the older flies prevent transposon RNA elevation in old age. In addition, they observed increased lifespan in these modified flies, an outcome that is akin to finding a great therapist who can talk you through issues and not letting the "extra baggage" like transposons continue to weigh you down.
Beyond the genome, the BUSM researchers also discovered that transposons could accumulate as extra-chromosomal circular DNAs. These mysterious circles are distinct from the genome and have also been seen to exist in cancer cells. Although genomic transposons only accumulate in mutant flies, these circles may accumulate even in normal flies.
Future efforts by the Lau lab will be to continue finding out how transposon circular DNAs amass during aging; and if the enhancement of RNA interference in older animals can also halt circular DNA in addition to transposon RNA. "Maybe we could all use some good therapy, not only for our emotional state but for our genomes as well," adds Lau.
More information: Nachen Yang et al, Transposable element landscapes in aging Drosophila, PLOS Genetics (2022). DOI: 10.1371/journal.pgen.1010024 , journals.plos.org/plosgenetics … journal.pgen.1010024
Journal information: PLoS Genetics
Provided by Boston University School of Medicine