Nano-sized vesicles with ACE2 receptor could prevent, treat infection from current and future strains of SARS-CoV-2
Scientists at The University of Texas MD Anderson Cancer Center and Northwestern Medicine have identified natural extracellular vesicles containing the ACE2 protein (evACE2) in the blood of COVID-19 patients that can block infection from broad strains of SARS-CoV-2 virus in preclinical studies. The study was published today in Nature Communications.
The evACE2 act as decoys in the body and can serve as a therapeutic to be developed for prevention and treatment for current and future strains of SARS-CoV-2 and subsequent coronaviruses, the scientists report. Once developed as a therapeutic product, evACE2 have the potential to benefit humans as a biological treatment with minimal toxicities.
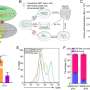
The study is the first to show evACE2 are capable of fighting the new SARS-CoV-2 variants with an equal or better efficacy than blocking the original strain. The researchers found that evACE2 exist in human blood as a natural anti-viral response. The more severe, the higher the levels of evACE2 detected in the patient's blood.
"Whenever a new mutant strain of SARS-CoV-2 surges, the original vaccine and therapeutic antibodies may lose power against alpha, beta, delta and the most recent, omicron," said co-senior author Huiping Liu, M.D., Ph.D., associate professor of pharmacology and medicine at Northwestern University Feinberg School of Medicine. "However, the beauty of evACE2 is its superpower in blocking broad strains of coronaviruses, including current SARS-CoV-2 and even future SARS coronaviruses from infecting humans. Our mouse studies demonstrate the therapeutic potential of evACE2 in preventing or blocking SARS-CoV-2 infection when it is delivered to the airway via droplets."
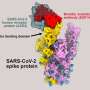
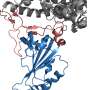
The evACE2 are tiny lipid bubbles in nanoparticle size that express the ACE2 protein, like handles for the virus to grab. These vesicles act as decoys to lure the SARS-CoV-2 virus away from the ACE2 protein on cells, which is how the virus infects cells. The virus spike protein grabs evACE2 instead of cellular ACE2, preventing it from entering the cell. Once captured, the virus will either float harmlessly around or be cleared by a macrophage immune cell. It can no longer cause infection.
"The key takeaway from this study is the identification of naturally occurring extracellular vesicles in the body that express the ACE2 receptor on their surface and serve as part of the normal adaptive defense against COVID-19-causing viruses," said co-senior author Raghu Kalluri, M.D., Ph.D., chair of Cancer Biology at MD Anderson. "Building upon this, we've discovered a way to harness this natural defense as a new potential therapy against this devastating virus."
The COVID-19 pandemic has been extended and challenged by a constantly changing SARS-CoV-2 virus. One of the biggest challenges is the moving target of pathogenic coronavirus that constantly evolves into new virus strains (variants) with mutations. These new viral strains harbor various changes in the viral protein spike with high infection rates and increased breakthroughs due to vaccine inefficiencies and resistance to therapeutic monoclonal antibodies.
"Our studies demonstrate that extracellular vesicles act to neutralize SARS-CoV-2 infection and highlight the potential for extracellular vesicles to play a broader role in defense against other types of infection which could be exploited therapeutically," said co-lead author Kathleen McAndrews, Ph.D., postdoctoral fellow in Cancer Biology at MD Anderson.
Northwestern and MD Anderson have a pending patent on evACE2. The goal is to collaborate with industry partners and develop evACE2 as a biological therapeutic product (nasal spray or injected therapeutics) for prevention and treatment of COVID-19. Liu and another co-senior author, Deyu Fang from pathology at Northwestern, have formed a startup company, Exomira, to take this patent and develop evACE2 as a therapeutic.
"It remains urgent to identify novel therapeutics," Liu said. "We think evACE2 can meet the challenges and fight against broad strains of SARS-CoV-2 and future emerging coronaviruses to protect the immunocompromised (at least 2.7% of U.S. adults), the unvaccinated (94% in low-income countries and more than 30% in the U.S.) and even the vaccinated from breakthrough infections."
A team of more than 30 authors collaborated on this work. They include four lead co-first authors Lamiaa El-Shennawy, Andrew Hoffmann and Nurmaa Dashzeveg, all from the Liu lab at Northwestern, and McAndrews from the Kalluri Lab of MD Anderson.
More information: Circulating ACE2-expressing extracellular vesicles block broad strains of SARS-CoV-2, Nature Communications (2022). DOI: 10.1038/s41467-021-27893-2 , www.nature.com/articles/s41467-021-27893-2
Journal information: Nature Communications
Provided by University of Texas M. D. Anderson Cancer Center